Investigating confounding in network-based test-negative design influenza vaccine effectiveness studies-Experience from the DRIVE project
- PMID: 36550627
- PMCID: PMC9835455
- DOI: 10.1111/irv.13087
Investigating confounding in network-based test-negative design influenza vaccine effectiveness studies-Experience from the DRIVE project
Abstract
Background: Establishing a large study network to conduct influenza vaccine effectiveness (IVE) studies while collecting appropriate variables to account for potential bias is important; the most relevant variables should be prioritized. We explored the impact of potential confounders on IVE in the DRIVE multi-country network of sites conducting test-negative design (TND) studies.
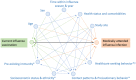
Methods: We constructed a directed acyclic graph (DAG) to map the relationship between influenza vaccination, medically attended influenza infection, confounders, and other variables. Additionally, we used the Development of Robust and Innovative Vaccines Effectiveness (DRIVE) data from the 2018/2019 and 2019/2020 seasons to explore the effect of covariate adjustment on IVE estimates. The reference model was adjusted for age, sex, calendar time, and season. The covariates studied were presence of at least one, two, or three chronic diseases; presence of six specific chronic diseases; and prior healthcare use. Analyses were conducted by site and subsequently pooled.
Results: The following variables were included in the DAG: age, sex, time within influenza season and year, health status and comorbidities, study site, health-care-seeking behavior, contact patterns and social precautionary behavior, socioeconomic status, and pre-existing immunity. Across all age groups and settings, only adjustment for lung disease in older adults in the primary care setting resulted in a relative change of the IVE point estimate >10%.
Conclusion: Our study supports a parsimonious approach to confounder adjustment in TND studies, limited to adjusting for age, sex, and calendar time. Practical implications are that necessitating fewer variables lowers the threshold for enrollment of sites in IVE studies and simplifies the pooling of data from different IVE studies or study networks.
Keywords: adjustment; confounders; covariate; influenza vaccine effectiveness; test-negative design.
© 2022 The Authors. Influenza and Other Respiratory Viruses published by John Wiley & Sons Ltd.
Conflict of interest statement
Anke Stuurman is an employee of P95. P95 holds/has held contracts with AstraZeneca, GSK, Sanofi, and Seqirus. Miriam Levi has no potential conflict of interest. Phillipe Beutels reports grants from the Innovative Medicines Initiative of the European Commission. He attended meetings of an advisory board on economic evaluations of vaccines convened by Pfizer in 2019, outside the submitted work. Hélène Bricout is a full‐time employee of Sanofi and holds shares in Sanofi. Alexandre Descamps reports consultation fees from Sanofi for advisory board meetings, outside the submitted work. Gael Dos Santos is an employee of the GSK group of companies and holds shares in the GSK group of companies as part of his annual remuneration. Ian McGovern is an employee of Seqirus and holds shares in Seqirus. Ainara Mira‐Iglesias has no potential conflict of interest. Jos Nauta is employed by Abbott, a company that produces influenza vaccines, and hold shares in Abbott. Laurence Torcel‐Pagnon is an employee of Sanofi and holds shares in Sanofi. Jorne Biccler is an employee of P95. P95 holds/has held contracts with AstraZeneca, GSK, Sanofi, and Seqirus.
Figures

References
-
- DRIVE consortium. DRIVE – Development of Robust and Innovative Vaccine Effectiveness: Increasing understanding of influenza vaccine effectiveness in Europe. 2022. https://www.drive-eu.org/
-
- Stuurman A, Bollaerts K, Alexandridou M, et al. D7.4 Setting up brand‐specific influenza vaccine effectiveness studies in Europe – results of the pilot season 2017/18. 2018. https://www.drive-eu.org/wp-content/uploads/2018/12/D7_4_Report-pilot-se...
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

