Hypothermic Preservation of Adipose-Derived Mesenchymal Stromal Cells as a Viable Solution for the Storage and Distribution of Cell Therapy Products
- PMID: 36551011
- PMCID: PMC9774331
- DOI: 10.3390/bioengineering9120805
Hypothermic Preservation of Adipose-Derived Mesenchymal Stromal Cells as a Viable Solution for the Storage and Distribution of Cell Therapy Products
Abstract
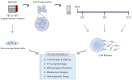
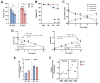
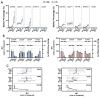
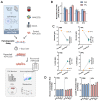
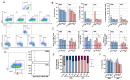
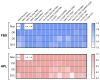
Cell and gene therapies (CGT) have reached new therapeutic targets but have noticeably high prices. Solutions to reduce production costs might be found in CGT storage and transportation since they typically involve cryopreservation, which is a heavily burdened process. Encapsulation at hypothermic temperatures (e.g., 2-8 °C) could be a feasible alternative. Adipose tissue-derived mesenchymal stromal cells (MSC(AT)) expanded using fetal bovine serum (FBS)- (MSC-FBS) or human platelet lysate (HPL)-supplemented mediums (MSC-HPL) were encapsulated in alginate beads for 30 min, 5 days, and 12 days. After bead release, cell recovery and viability were determined to assess encapsulation performance. MSC identity was verified by flow cytometry, and a set of assays was performed to evaluate functionality. MSC(AT) were able to survive encapsulated for a standard transportation period of 5 days, with recovery values of 56 ± 5% for MSC-FBS and 77 ± 6% for MSC-HPL (which is a negligible drop compared to earlier timepoints). Importantly, MSC function did not suffer from encapsulation, with recovered cells showing robust differentiation potential, expression of immunomodulatory molecules, and hematopoietic support capacity. MSC(AT) encapsulation was proven possible for a remarkable 12 day period. There is currently no solution to completely replace cryopreservation in CGT logistics and supply chain, although encapsulation has shown potential to act as a serious competitor.
Keywords: cell encapsulation; fetal bovine serum; hematopoietic support assay; human platelet lysate; hypothermic temperatures; mesenchymal stromal cells; xenogeneic-free.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Influence of the mesenchymal stromal cell source on the hematopoietic supportive capacity of umbilical cord blood-derived CD34+-enriched cells.Stem Cell Res Ther. 2021 Jul 13;12(1):399. doi: 10.1186/s13287-021-02474-8. Stem Cell Res Ther. 2021. PMID: 34256848 Free PMC article.
-
Human platelet lysate stimulates high-passage and senescent human multipotent mesenchymal stromal cell growth and rejuvenation in vitro.Cytotherapy. 2013 Dec;15(12):1469-83. doi: 10.1016/j.jcyt.2013.05.020. Epub 2013 Aug 24. Cytotherapy. 2013. PMID: 23981539
-
Reconstitution and post-thaw storage of cryopreserved human mesenchymal stromal cells: Pitfalls and optimizations for clinically compatible formulants.Regen Ther. 2023 Apr 20;23:67-75. doi: 10.1016/j.reth.2023.03.006. eCollection 2023 Jun. Regen Ther. 2023. PMID: 37153832 Free PMC article.
-
Systematic review and meta-analysis on the use of human platelet lysate for mesenchymal stem cell cultures: comparison with fetal bovine serum and considerations on the production protocol.Stem Cell Res Ther. 2022 Apr 4;13(1):142. doi: 10.1186/s13287-022-02815-1. Stem Cell Res Ther. 2022. PMID: 35379348 Free PMC article.
-
Human platelet lysate to substitute fetal bovine serum in hMSC expansion for translational applications: a systematic review.J Transl Med. 2020 Sep 15;18(1):351. doi: 10.1186/s12967-020-02489-4. J Transl Med. 2020. PMID: 32933520 Free PMC article.
Cited by
-
Bioinks for Space Missions: The Influence of Long-Term Storage of Alginate-Methylcellulose-Based Bioinks on Printability as well as Cell Viability and Function.Adv Healthc Mater. 2023 Sep;12(23):e2300436. doi: 10.1002/adhm.202300436. Epub 2023 May 12. Adv Healthc Mater. 2023. PMID: 37125819 Free PMC article.
-
Advances in ex vivo expansion of hematopoietic stem and progenitor cells for clinical applications.Front Bioeng Biotechnol. 2024 May 23;12:1380950. doi: 10.3389/fbioe.2024.1380950. eCollection 2024. Front Bioeng Biotechnol. 2024. PMID: 38846805 Free PMC article. Review.
-
"Remodeling the intestinal immune microenvironment": immune regulation and tissue regeneration by mesenchymal stem/stromal cells in the repair microenvironment of inflammatory bowel disease.Front Immunol. 2025 May 13;16:1543702. doi: 10.3389/fimmu.2025.1543702. eCollection 2025. Front Immunol. 2025. PMID: 40433382 Free PMC article. Review.
References
-
- Committee for Medicinal Products for Human Use . Alofisel Assessment Report. CHMP; London, UK: 2017.
Grants and funding
- LA/P/0140/2020/Fundação para a Ciência e Tecnologia
- PTDC/EQU-EQU/31651/2017/Fundação para a Ciência e Tecnologia
- UIDB/04565/2020 and UIDP/04565/2020/Fundação para a Ciência e Tecnologia
- POCI-01-0247-FEDER-038313/Programa Operacional Competitividade e Internacionalização and European Regional Development Fund
LinkOut - more resources
Full Text Sources
Research Materials

