Review: Advanced Atomic Force Microscopy Modes for Biomedical Research
- PMID: 36551083
- PMCID: PMC9775674
- DOI: 10.3390/bios12121116
Review: Advanced Atomic Force Microscopy Modes for Biomedical Research
Abstract
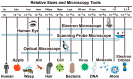
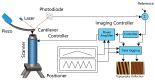
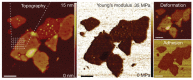
Visualization of biomedical samples in their native environments at the microscopic scale is crucial for studying fundamental principles and discovering biomedical systems with complex interaction. The study of dynamic biological processes requires a microscope system with multiple modalities, high spatial/temporal resolution, large imaging ranges, versatile imaging environments and ideally in-situ manipulation capabilities. Recent development of new Atomic Force Microscopy (AFM) capabilities has made it such a powerful tool for biological and biomedical research. This review introduces novel AFM functionalities including high-speed imaging for dynamic process visualization, mechanobiology with force spectroscopy, molecular species characterization, and AFM nano-manipulation. These capabilities enable many new possibilities for novel scientific research and allow scientists to observe and explore processes at the nanoscale like never before. Selected application examples from recent studies are provided to demonstrate the effectiveness of these AFM techniques.
Keywords: atomic force microscopy; biomedical research; high-speed imaging; material property mapping; mechanobiology; nano-manipulation; nanotechnology.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
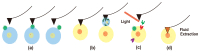


References
-
- Mou J., Czajkowsky D.M., Zhang Y., Shao Z. High-resolution atomic-force microscopy of DNA: The pitch of the double helix. FEBS Lett. 1995;371:279–282. - PubMed
-
- Miyata K., Tracey J., Miyazawa K., Haapasilta V., Spijker P., Kawagoe Y., Foster A.S., Tsukamoto K., Fukuma T. Dissolution processes at step edges of calcite in water investigated by high-speed frequency modulation atomic force microscopy and simulation. Nano Lett. 2017;17:4083–4089. doi: 10.1021/acs.nanolett.7b00757. - DOI - PubMed
-
- Xia F., Quigley J., Zhang X., Yang C., Wang Y., Youcef-Toumi K. A modular low-cost atomic force microscope for precision mechatronics education. Mechatronics. 2021;76:102550. doi: 10.1016/j.mechatronics.2021.102550. - DOI
-
- Rangelow I.W., Ivanov T., Ahmad A., Kaestner M., Lenk C., Bozchalooi I.S., Xia F., Youcef-Toumi K., Holz M., Reum A. Review Article: Active scanning probes: A versatile toolkit for fast imaging and emerging nanofabrication. J. Vac. Sci. Technol. B. 2017;35:06G101. doi: 10.1116/1.4992073. - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

