Increasing the Efficacy of Treatment of Staphylococcus aureus- Candida albicans Mixed Infections with Myrtenol
- PMID: 36551400
- PMCID: PMC9774912
- DOI: 10.3390/antibiotics11121743
Increasing the Efficacy of Treatment of Staphylococcus aureus- Candida albicans Mixed Infections with Myrtenol
Abstract
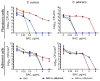
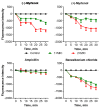
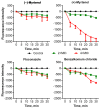
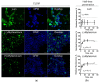
Infectious diseases caused by various nosocomial microorganisms affect worldwide both immunocompromised and relatively healthy persons. Bacteria and fungi have different tools to evade antimicrobials, such as hydrolysis damaging the drug, efflux systems, and the formation of biofilm that significantly complicates the treatment of the infection. Here, we show that myrtenol potentiates the antimicrobial and biofilm-preventing activity of conventional drugs against S. aureus and C. albicans mono- and dual-species cultures. In our study, the two optical isomers, (-)-myrtenol and (+)-myrtenol, have been tested as either antibacterials, antifungals, or enhancers of conventional drugs. (+)-Myrtenol demonstrated a synergistic effect with amikacin, fluconazole, and benzalkonium chloride on 64-81% of the clinical isolates of S. aureus and C. albicans, including MRSA and fluconazole-resistant fungi, while (-)-myrtenol increased the properties of amikacin and fluconazole to repress biofilm formation in half of the S. aureus and C. albicans isolates. Furthermore, myrtenol was able to potentiate benzalkonium chloride up to sixteen-fold against planktonic cells in an S. aureus-C. albicans mixed culture and repressed the adhesion of S. aureus. The mechanism of both (-)-myrtenol and (+)-myrtenol synergy with conventional drugs was apparently driven by membrane damage since the treatment with both terpenes led to a significant drop in membrane potential similar to the action of benzalkonium chloride. Thus, due to the low toxicity of myrtenol, it seems to be a promising agent to increase the efficiency of the treatment of infections caused by bacteria and be fungi of the genus Candida as well as mixed fungal-bacterial infections, including resistant strains.
Keywords: Candida albicans; Staphylcoccus aureus; benzalkonium chloride; drug synergism; mixed infections; myrtenol.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Antimicrobial and Biofilm-Preventing Activity of l-Borneol Possessing 2(5H)-Furanone Derivative F131 against S. aureus—C. albicans Mixed Cultures.Pathogens. 2022 Dec 23;12(1):26. doi: 10.3390/pathogens12010026. Pathogens. 2022. PMID: 36678375 Free PMC article.
-
Antibiofilm efficacy of the gold compound auranofin on dual species biofilms of Staphylococcus aureus and Candida sp.J Appl Microbiol. 2020 Jan;128(1):88-101. doi: 10.1111/jam.14443. Epub 2019 Nov 19. J Appl Microbiol. 2020. PMID: 31509623
-
Exploring the Antimicrobial Potential of a Peptide Against Mixed Biofilm of Staphylococcus aureus and Candida albicans.Probiotics Antimicrob Proteins. 2025 Jun 10. doi: 10.1007/s12602-025-10571-w. Online ahead of print. Probiotics Antimicrob Proteins. 2025. PMID: 40493322
-
Candida and candidaemia. Susceptibility and epidemiology.Dan Med J. 2013 Nov;60(11):B4698. Dan Med J. 2013. PMID: 24192246 Review.
-
Metabolic Adaptations During Staphylococcus aureus and Candida albicans Co-Infection.Front Immunol. 2021 Dec 8;12:797550. doi: 10.3389/fimmu.2021.797550. eCollection 2021. Front Immunol. 2021. PMID: 34956233 Free PMC article. Review.
Cited by
-
Preparation and analysis of quinoa active protein (QAP) and its mechanism of inhibiting Candida albicans from a transcriptome perspective.PeerJ. 2025 Feb 14;13:e18961. doi: 10.7717/peerj.18961. eCollection 2025. PeerJ. 2025. PMID: 39963196 Free PMC article.
-
Monoterpene Thiols: Synthesis and Modifications for Obtaining Biologically Active Substances.Int J Mol Sci. 2023 Nov 1;24(21):15884. doi: 10.3390/ijms242115884. Int J Mol Sci. 2023. PMID: 37958865 Free PMC article. Review.
-
Advances in Human Pathogen Control-A 21st Century Challenge.Vaccines (Basel). 2023 Sep 2;11(9):1449. doi: 10.3390/vaccines11091449. Vaccines (Basel). 2023. PMID: 37766126 Free PMC article.
-
Myrtenol's Effectiveness against Hospital-Acquired Methicillin-Resistant Staphylococcus aureus: Targeting Antibiofilm and Antivirulence Properties.Int J Microbiol. 2024 Oct 16;2024:8832448. doi: 10.1155/2024/8832448. eCollection 2024. Int J Microbiol. 2024. PMID: 39445102 Free PMC article.
-
The Antimicrobial Potential of the Hop (Humulus lupulus L.) Extract against Staphylococcus aureus and Oral Streptococci.Pharmaceuticals (Basel). 2024 Jan 27;17(2):162. doi: 10.3390/ph17020162. Pharmaceuticals (Basel). 2024. PMID: 38399377 Free PMC article.
References
-
- Khan H.A., Baig F.K., Mehboob R. Nosocomial infections: Epidemiology, prevention, control and surveillance. Asian Pac. J. Trop. Biomed. 2017;7:478–482. doi: 10.1016/j.apjtb.2017.01.019. - DOI
-
- Petrosillo N., Pagani L., Ippolito G. Nosocomial infections in HIV-positive patients: An overview. Infection. 2003;31:28–34. - PubMed
-
- Bardi T., Pintado V., Gomez-Rojo M., Escudero-Sanchez R., Azzam Lopez A., Diez-Remesal Y., Martinez Castro N., Ruiz-Garbajosa P., Pestaña D. Nosocomial infections associated to COVID-19 in the intensive care unit: Clinical characteristics and outcome. Eur. J. Clin. Microbiol. Infect. Dis. 2021;40:495–502. doi: 10.1007/s10096-020-04142-w. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

