Strategies to Mitigate and Treat Orthopaedic Device-Associated Infections
- PMID: 36551479
- PMCID: PMC9774155
- DOI: 10.3390/antibiotics11121822
Strategies to Mitigate and Treat Orthopaedic Device-Associated Infections
Abstract
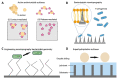
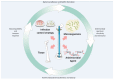
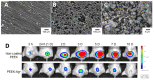
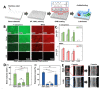
Orthopaedic device implants play a crucial role in restoring functionality to patients suffering from debilitating musculoskeletal diseases or to those who have experienced traumatic injury. However, the surgical implantation of these devices carries a risk of infection, which represents a significant burden for patients and healthcare providers. This review delineates the pathogenesis of orthopaedic implant infections and the challenges that arise due to biofilm formation and the implications for treatment. It focuses on research advancements in the development of next-generation orthopaedic medical devices to mitigate against implant-related infections. Key considerations impacting the development of devices, which must often perform multiple biological and mechanical roles, are delineated. We review technologies designed to exert spatial and temporal control over antimicrobial presentation and the use of antimicrobial surfaces with intrinsic antibacterial activity. A range of measures to control bio-interfacial interactions including approaches that modify implant surface chemistry or topography to reduce the capacity of bacteria to colonise the surface, form biofilms and cause infections at the device interface and surrounding tissues are also reviewed.
Keywords: antimicrobial; biofilm; bioinspired; drug delivery; implant coating; infection; medical device; nanotechnology; orthopaedic implants; polymer.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures




References
-
- McMillan D.J., Lutton C., Rosenzweig N., Sriprakash K.S., Goss B., Stemberger M., Schuetz M.A., Steck R. Prevention of staphylococcus aures biofilm formation on metallic surgical implants via controlled release of gentamicin. J. Biomed. Sci. Eng. 2011;4:535–542. doi: 10.4236/jbise.2011.48069. - DOI
-
- Evans N.T., Torstrick F.B., Lee C.S.D., Dupont K.M., Safranski D.L., Chang W.A., Macedo A.E., Lin A.S.P., Boothby J.M., Whittingslow D.C., et al. High-strength, surface-porous polyether-ether-ketone for load-bearing orthopedic implants. Acta Biomater. 2015;13:159–167. doi: 10.1016/j.actbio.2014.11.030. - DOI - PMC - PubMed
-
- O’ Sullivan C., Kennedy G., O’ Neill L., Crean A.M., Ryan K.B. Biomedical Applications of Inorganic Materials. The Royal Society of Chemistry; Cambridge, UK: 2022. Chapter 5 Inorganic Biomaterials to Support the Formation and Repair of Bone Tissue; pp. 242–304.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

