GPX2 Gene Affects Feed Efficiency of Pigs by Inhibiting Fat Deposition and Promoting Muscle Development
- PMID: 36552449
- PMCID: PMC9774625
- DOI: 10.3390/ani12243528
GPX2 Gene Affects Feed Efficiency of Pigs by Inhibiting Fat Deposition and Promoting Muscle Development
Abstract
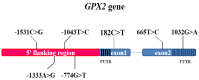
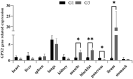
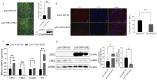
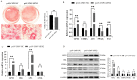
GPX2 has been recognized as a potential candidate gene for feed efficiency in pigs. This article aimed to elucidate polymorphism of GPX2 associated with feed efficiency and its related molecular mechanism. In this study, seven single nucleotide polymorphisms (SNP) of GPX2 were found among 383 Duroc pigs. In addition, seven SNPs and ALGA0043483 (PorcineSNP60 BeadChip data in 600 Duroc pigs), which are near the GPX2 gene, were identified in one haplotypes block. Furthermore, associated studies showed that the genotype of GPX2 has significant association with weaning weight and 100 kg BF in Duroc pigs. In addition, the AG had no effect when the backfat became thinner, and the FCR and RFI traits had a tendency to decrease in the G3 + TT combination genotype, accompanied by an increase of GPX2 expression in backfat and muscle tissues. At the cellular level, the adipocyte proliferation and ability of adipogenic differentiation were reduced, and the lipid degradation increased in 3T3-L1 when there was overexpression of GPX2. In contrast, overexpression of the GPX2 gene can promote the muscle cell proliferation and myogenic differentiation in C2C12 cells. In other words, GPX2 has the effect of reducing fat deposition and promoting muscle development, and it is a candidate gene for backfat and feed efficiency.
Keywords: GPX2; SNP; adipocyte; backfat; development; feed efficiency; molecular mechanism; muscle cells.
Conflict of interest statement
We certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript. L.P., Z.W., Y.L., Y.D., C.Z. (Chunting Zheng), X.Z., L.Q., C.Z. (Chunguang Zhang), H.L., J.Z., L.G. and L.W. declared no competing interests.
Figures




Similar articles
-
Overexpression of GPX2 gene regulates the development of porcine preadipocytes and skeletal muscle cells through MAPK signaling pathway.PLoS One. 2024 May 9;19(5):e0298827. doi: 10.1371/journal.pone.0298827. eCollection 2024. PLoS One. 2024. PMID: 38722949 Free PMC article.
-
Genome-wide association and systems genetic analyses of residual feed intake, daily feed consumption, backfat and weight gain in pigs.BMC Genet. 2014 Feb 17;15:27. doi: 10.1186/1471-2156-15-27. BMC Genet. 2014. PMID: 24533460 Free PMC article.
-
Feed intake, average daily gain, feed efficiency, and real-time ultrasound traits in Duroc pigs: I. Genetic parameter estimation and accuracy of genomic prediction.J Anim Sci. 2014 Jun;92(6):2377-86. doi: 10.2527/jas.2013-7338. Epub 2014 Mar 26. J Anim Sci. 2014. PMID: 24671579
-
Genome-wide association studies for seven production traits highlight genomic regions useful to dissect dry-cured ham quality and production traits in Duroc heavy pigs.Animal. 2018 Sep;12(9):1777-1784. doi: 10.1017/S1751731118000757. Epub 2018 Apr 30. Animal. 2018. PMID: 29706143
-
Feed intake, average daily gain, feed efficiency, and real-time ultrasound traits in Duroc pigs: II. Genomewide association.J Anim Sci. 2014 Jul;92(7):2846-60. doi: 10.2527/jas.2014-7337. J Anim Sci. 2014. PMID: 24962532
Cited by
-
Overexpression of GPX2 gene regulates the development of porcine preadipocytes and skeletal muscle cells through MAPK signaling pathway.PLoS One. 2024 May 9;19(5):e0298827. doi: 10.1371/journal.pone.0298827. eCollection 2024. PLoS One. 2024. PMID: 38722949 Free PMC article.
-
Mechanism of Myh3 gene regulation of intramuscular fat content in Beijing black pigs via the MAPK signaling pathway.BMC Genomics. 2025 Jul 7;26(1):642. doi: 10.1186/s12864-025-11528-7. BMC Genomics. 2025. PMID: 40624626 Free PMC article.
-
Regulatory Effects of 198-bp Structural Variants in the GSTA2 Promoter Region on Adipogenesis in Chickens.Int J Mol Sci. 2024 Jun 28;25(13):7155. doi: 10.3390/ijms25137155. Int J Mol Sci. 2024. PMID: 39000259 Free PMC article.
-
STC2 Inhibits Hepatic Lipid Synthesis and Correlates with Intramuscular Fatty Acid Composition, Body Weight and Carcass Traits in Chickens.Animals (Basel). 2024 Jan 25;14(3):383. doi: 10.3390/ani14030383. Animals (Basel). 2024. PMID: 38338026 Free PMC article.
References
-
- Huang J., Jia Y., Li Q., Son K., Hamilton C., Burris W.R., Bridges P.J., Stromberg A.J., Matthews J.C. Glutathione content and expression of proteins involved with glutathione metabolism differs in longissimus dorsi, subcutaneous adipose, and liver tissues of finished vs. growing beef steers. J. Anim. Sci. 2018;96:5152–5165. doi: 10.1093/jas/sky362. - DOI - PMC - PubMed
Grants and funding
- 2021-QY-204/Key R&D and Transformation Program of Qinghai Province Science and Technology Assistance Program for Qinghai, China
- 20JCQNJC00650/Tianjin Natural Science Foundation, China
- 2018KJ188/Scientific Research Program of Tianjin Education Commission, China
- ITTPRS2021006/Tianjin pig industry technology system innovation team, China
LinkOut - more resources
Full Text Sources
Miscellaneous

