Membrane Lipid Reshaping Underlies Oxidative Stress Sensing by the Mitochondrial Proteins UCP1 and ANT1
- PMID: 36552523
- PMCID: PMC9774536
- DOI: 10.3390/antiox11122314
Membrane Lipid Reshaping Underlies Oxidative Stress Sensing by the Mitochondrial Proteins UCP1 and ANT1
Abstract
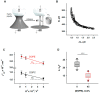
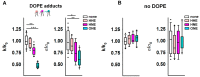
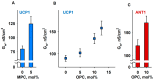
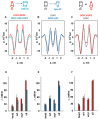
Oxidative stress and ROS are important players in the pathogenesis of numerous diseases. In addition to directly altering proteins, ROS also affects lipids with negative intrinsic curvature such as phosphatidylethanolamine (PE), producing PE adducts and lysolipids. The formation of PE adducts potentiates the protonophoric activity of mitochondrial uncoupling proteins, but the molecular mechanism remains unclear. Here, we linked the ROS-mediated change in lipid shape to the mechanical properties of the membrane and the function of uncoupling protein 1 (UCP1) and adenine nucleotide translocase 1 (ANT1). We show that the increase in the protonophoric activity of both proteins occurs due to the decrease in bending modulus in lipid bilayers in the presence of lysophosphatidylcholines (OPC and MPC) and PE adducts. Moreover, MD simulations showed that modified PEs and lysolipids change the lateral pressure profile of the membrane in the same direction and by the similar amplitude, indicating that modified PEs act as lipids with positive intrinsic curvature. Both results indicate that oxidative stress decreases stored curvature elastic stress (SCES) in the lipid bilayer membrane. We demonstrated that UCP1 and ANT1 sense SCES and proposed a novel regulatory mechanism for the function of these proteins. The new findings should draw the attention of the scientific community to this important and unexplored area of redox biochemistry.
Keywords: bending moduli; lateral pressure profile; lipid shape; lipid–protein interaction; mitochondrial membrane protein; protonophoric function; reactive aldehydes; stored curvature elastic stress.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures


Similar articles
-
Mitochondrial Uncoupling Proteins (UCP1-UCP3) and Adenine Nucleotide Translocase (ANT1) Enhance the Protonophoric Action of 2,4-Dinitrophenol in Mitochondria and Planar Bilayer Membranes.Biomolecules. 2021 Aug 9;11(8):1178. doi: 10.3390/biom11081178. Biomolecules. 2021. PMID: 34439844 Free PMC article.
-
The molecular mechanism behind reactive aldehyde action on transmembrane translocations of proton and potassium ions.Free Radic Biol Med. 2015 Dec;89:1067-76. doi: 10.1016/j.freeradbiomed.2015.10.422. Epub 2015 Oct 28. Free Radic Biol Med. 2015. PMID: 26520807 Free PMC article.
-
Transgenic overexpression of adenine nucleotide translocase 1 protects ischemic hearts against oxidative stress.J Mol Med (Berl). 2016 Jun;94(6):645-53. doi: 10.1007/s00109-016-1413-4. Epub 2016 Apr 15. J Mol Med (Berl). 2016. PMID: 27080394
-
The Role of Phosphatidylethanolamine Adducts in Modification of the Activity of Membrane Proteins under Oxidative Stress.Molecules. 2019 Dec 12;24(24):4545. doi: 10.3390/molecules24244545. Molecules. 2019. PMID: 31842328 Free PMC article. Review.
-
Does membrane curvature elastic energy play a role in mediating oxidative stress in lipid membranes?Free Radic Biol Med. 2021 Aug 1;171:191-202. doi: 10.1016/j.freeradbiomed.2021.05.021. Epub 2021 May 15. Free Radic Biol Med. 2021. PMID: 34000382 Review.
Cited by
-
Exploring the proton transport mechanism of the mitochondrial ADP/ATP carrier: FA-cycling hypothesis and beyond.Protein Sci. 2025 Mar;34(3):e70047. doi: 10.1002/pro.70047. Protein Sci. 2025. PMID: 39969060 Free PMC article. Review.
-
Investigating How Lysophosphatidylcholine and Lysophosphatidylethanolamine Enhance the Membrane Permeabilization Efficacy of Host Defense Peptide Piscidin 1.J Phys Chem B. 2025 Jan 9;129(1):210-227. doi: 10.1021/acs.jpcb.4c05845. Epub 2024 Dec 16. J Phys Chem B. 2025. PMID: 39681296
-
Molecular Dynamics Simulations of a Putative Novel Mechanism for UCP1-Assisted FA Anion Transport.Acta Physiol (Oxf). 2025 Jul;241(7):e70068. doi: 10.1111/apha.70068. Acta Physiol (Oxf). 2025. PMID: 40497434 Free PMC article.
-
Mitochondrial phospholipid metabolism in health and disease.J Cell Sci. 2023 Sep 1;136(17):jcs260857. doi: 10.1242/jcs.260857. Epub 2023 Sep 1. J Cell Sci. 2023. PMID: 37655851 Free PMC article.
References
-
- Sam P.N., Calzada E., Acoba M.G., Zhao T., Watanabe Y., Nejatfard A., Trinidad J.C., Shutt T.E., Neal S.E., Claypool S.M. Impaired phosphatidylethanolamine metabolism activates a reversible stress response that detects and resolves mutant mitochondrial precursors. iScience. 2021;24:102196. doi: 10.1016/j.isci.2021.102196. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

