Nrf2 and Oxidative Stress: A General Overview of Mechanisms and Implications in Human Disease
- PMID: 36552553
- PMCID: PMC9774434
- DOI: 10.3390/antiox11122345
Nrf2 and Oxidative Stress: A General Overview of Mechanisms and Implications in Human Disease
Abstract
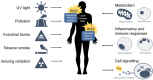
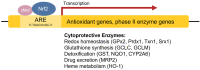
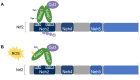
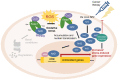
Organisms are continually exposed to exogenous and endogenous sources of reactive oxygen species (ROS) and other oxidants that have both beneficial and deleterious effects on the cell. ROS have important roles in a wide range of physiological processes; however, high ROS levels are associated with oxidative stress and disease progression. Oxidative stress has been implicated in nearly all major human diseases, from neurogenerative diseases and neuropsychiatric disorders to cardiovascular disease, diabetes, and cancer. Antioxidant defence systems have evolved as a means of protection against oxidative stress, with the transcription factor Nrf2 as the key regulator. Nrf2 is responsible for regulating an extensive panel of antioxidant enzymes involved in the detoxification and elimination of oxidative stress and has been extensively studied in the disease contexts. This review aims to provide the reader with a general overview of oxidative stress and Nrf2, including basic mechanisms of Nrf2 activation and regulation, and implications in various major human diseases.
Keywords: Nrf2; antioxidant; antioxidant response; oxidative stress.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Halliwell B., Gutteridge J.M.C. Free Radicals in Biology and Medicine. 5th ed. Volume 944 Oxford University Press; Oxford, UK: 2015.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

