Oxidative Stress-Induced Overactivation of Frog Eggs Triggers Calcium-Dependent Non-Apoptotic Cell Death
- PMID: 36552641
- PMCID: PMC9774297
- DOI: 10.3390/antiox11122433
Oxidative Stress-Induced Overactivation of Frog Eggs Triggers Calcium-Dependent Non-Apoptotic Cell Death
Abstract
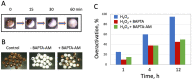
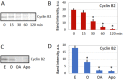
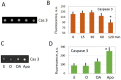
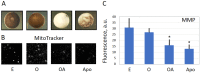
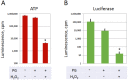
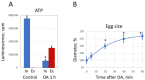
Excessive activation of frog eggs (overactivation) is a pathological process that renders eggs unfertilizable. Its physiological inducers are unknown. Previously, oxidative stress was shown to cause time- and dose-dependent overactivation of Xenopus laevis frog eggs. Here, we demonstrate that the oxidative stress-induced egg overactivation is a calcium-dependent phenomenon which can be attenuated in the presence of the selective calcium chelator BAPTA. Degradation of cyclin B2, which is known to be initiated by calcium transient in fertilized or parthenogenetically activated eggs, can also be observed in the overactivated eggs. Decline in mitochondrial membrane potential, ATP depletion and termination of protein synthesis manifest in the eggs within one hour of triggering overactivation. These intracellular events occur in the absence of caspase activation. Furthermore, plasma membrane integrity is compromised in the overactivated eggs, as evidenced by ATP leakage and egg swelling. In sum, our data demonstrate that oxidative stress-induced overactivation of frog eggs causes fast and dramatic disruption of cellular homeostasis, resulting in robust and expedited cell death by a calcium-dependent non-apoptotic mechanism.
Keywords: Xenopus laevis; cell death; eggs; overactivation; oxidative stress.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Egg Overactivation-An Overlooked Phenomenon of Gamete Physiology.Int J Mol Sci. 2025 Apr 27;26(9):4163. doi: 10.3390/ijms26094163. Int J Mol Sci. 2025. PMID: 40362401 Free PMC article. Review.
-
Spontaneous Overactivation of Xenopus Frog Eggs Triggers Necrotic Cell Death.Int J Mol Sci. 2024 May 13;25(10):5321. doi: 10.3390/ijms25105321. Int J Mol Sci. 2024. PMID: 38791359 Free PMC article.
-
Biochemical Hallmarks of Oxidative Stress-Induced Overactivation of Xenopus Eggs.Biomed Res Int. 2019 Jul 2;2019:7180540. doi: 10.1155/2019/7180540. eCollection 2019. Biomed Res Int. 2019. PMID: 31341903 Free PMC article.
-
Unfertilized frog eggs die by apoptosis following meiotic exit.BMC Cell Biol. 2011 Dec 23;12:56. doi: 10.1186/1471-2121-12-56. BMC Cell Biol. 2011. PMID: 22195698 Free PMC article.
-
Calcium signaling and meiotic exit at fertilization in Xenopus egg.Int J Mol Sci. 2014 Oct 15;15(10):18659-76. doi: 10.3390/ijms151018659. Int J Mol Sci. 2014. PMID: 25322156 Free PMC article. Review.
Cited by
-
Egg Overactivation-An Overlooked Phenomenon of Gamete Physiology.Int J Mol Sci. 2025 Apr 27;26(9):4163. doi: 10.3390/ijms26094163. Int J Mol Sci. 2025. PMID: 40362401 Free PMC article. Review.
-
Hydrogen Peroxide Signaling in Physiology and Pathology.Antioxidants (Basel). 2023 Mar 7;12(3):661. doi: 10.3390/antiox12030661. Antioxidants (Basel). 2023. PMID: 36978909 Free PMC article.
-
Spontaneous Overactivation of Xenopus Frog Eggs Triggers Necrotic Cell Death.Int J Mol Sci. 2024 May 13;25(10):5321. doi: 10.3390/ijms25105321. Int J Mol Sci. 2024. PMID: 38791359 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources

