Resveratrol: Its Path from Isolation to Therapeutic Action in Eye Diseases
- PMID: 36552655
- PMCID: PMC9774148
- DOI: 10.3390/antiox11122447
Resveratrol: Its Path from Isolation to Therapeutic Action in Eye Diseases
Abstract
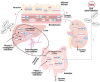
Due to the confirmed therapeutic potential of resveratrol (Rv) for eye diseases, namely its powerful anti-angiogenic and antioxidant effects, this molecule must be studied more deeply. Nowadays, the pharmaceutic and pharmacokinetic available studies offer a troubling picture because of its low stability and bioavailability. To overcome this problem, researchers started to design and create different delivery systems that could improve the delivery amount of Rv. Therefore, this review aims to shed light on the proper and efficient techniques to isolate, purify and quantify the Rv molecule, and how this therapeutic molecule can be a part of a delivery system. The Rv great impact on aspects regarding its stability, bioavailability and absorption are also debated here, based on the existent literature on in vitro and in vivo human and animal studies. Moreover, after its absorption the Rv influence at the molecular level in ocular pathologies is described. In addition, the present review summarizes the available literature about Rv, hoping that Rv will gain more attention to investigate its unexplored side.
Keywords: VEGF; delivery systems; ocular pathologies; resveratrol; sirtuins.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Resveratrol-delivery vehicle with anti-VEGF activity carried to human retinal pigmented epithelial cells exposed to high-glucose induced conditions.Colloids Surf B Biointerfaces. 2019 Sep 1;181:66-75. doi: 10.1016/j.colsurfb.2019.04.022. Epub 2019 May 7. Colloids Surf B Biointerfaces. 2019. PMID: 31125919
-
Health Effects of Resveratrol: Results from Human Intervention Trials.Nutrients. 2018 Dec 3;10(12):1892. doi: 10.3390/nu10121892. Nutrients. 2018. PMID: 30513922 Free PMC article. Review.
-
Resveratrol (RV): A pharmacological review and call for further research.Biomed Pharmacother. 2021 Nov;143:112164. doi: 10.1016/j.biopha.2021.112164. Epub 2021 Oct 2. Biomed Pharmacother. 2021. PMID: 34649335 Review.
-
Observation of human retinal remodeling in octogenarians with a resveratrol based nutritional supplement.Nutrients. 2013 Jun 4;5(6):1989-2005. doi: 10.3390/nu5061989. Nutrients. 2013. PMID: 23736827 Free PMC article.
-
Research Problems Associated with Resveratrol (trans-3, 5, 4'- trihydroxystilbene; RSV) and Various Strategies to Overcome those Problems (Review).Curr Drug Deliv. 2017;14(3):364-376. doi: 10.2174/1567201814666161109115735. Curr Drug Deliv. 2017. PMID: 27834152 Review.
Cited by
-
Formulating Resveratrol and Melatonin Self-Nanoemulsifying Drug Delivery Systems (SNEDDS) for Ocular Administration Using Design of Experiments.Pharmaceutics. 2024 Jan 18;16(1):125. doi: 10.3390/pharmaceutics16010125. Pharmaceutics. 2024. PMID: 38258134 Free PMC article.
-
One-year outcomes of resveratrol supplement with aflibercept versus aflibercept monotherapy in wet age-related macular degeneration.Int J Ophthalmol. 2023 Sep 18;16(9):1496-1502. doi: 10.18240/ijo.2023.09.17. eCollection 2023. Int J Ophthalmol. 2023. PMID: 37724273 Free PMC article.
-
Natural Product-Derived Senotherapeutics: Extraction and Biological Evaluation Techniques.Methods Mol Biol. 2025;2906:315-359. doi: 10.1007/978-1-0716-4426-3_19. Methods Mol Biol. 2025. PMID: 40082365
-
Resveratrol as a Novel Therapeutic Approach for Diabetic Retinopathy: Molecular Mechanisms, Clinical Potential, and Future Challenges.Molecules. 2025 Aug 4;30(15):3262. doi: 10.3390/molecules30153262. Molecules. 2025. PMID: 40807437 Free PMC article. Review.
-
Resveratrol Supplementation in Wet AMD: Association With Fewer Intravitreal Injections and Reduced Macular Fibrosis.Clin Ophthalmol. 2025 Jan 21;19:217-225. doi: 10.2147/OPTH.S494595. eCollection 2025. Clin Ophthalmol. 2025. PMID: 39867345 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

