Repair of Long Nerve Defects with a New Decellularized Nerve Graft in Rats and in Sheep
- PMID: 36552838
- PMCID: PMC9777287
- DOI: 10.3390/cells11244074
Repair of Long Nerve Defects with a New Decellularized Nerve Graft in Rats and in Sheep
Abstract
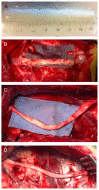
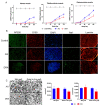
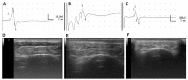
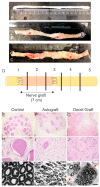
Decellularized nerve allografts (DC) are an alternative to autografts (AG) for repairing severe peripheral nerve injuries. We have assessed a new DC provided by VERIGRAFT. The decellularization procedure completely removed cellularity while preserving the extracellular matrix. We first assessed the DC in a 15 mm gap in the sciatic nerve of rats, showing slightly delayed but effective regeneration. Then, we assayed the DC in a 70 mm gap in the peroneal nerve of sheep compared with AG. Evaluation of nerve regeneration and functional recovery was performed by clinical, electrophysiology and ultrasound tests. No significant differences were found in functional recovery between groups of sheep. Histology showed a preserved fascicular structure in the AG while in the DC grafts regenerated axons were grouped in small units. In conclusion, the DC was permissive for axonal regeneration and allowed to repair a 70 mm long gap in the sheep nerve.
Keywords: allograft; autograft; decellularization; nerve regeneration; sheep.
Conflict of interest statement
E.C., S.T., S.B., J.F., E.JC., I.D.M., E.U. and X.N. declare that they have no competing financial interests. R.S. and S.H. are employees of VERIGRAFT AB.
Figures



References
-
- Pfister B.J., Gordon T., Loverde J.R., Kochar A.S., Mackinnon S.E., Cullen D.K. Biomedical Engineering Strategies for Peripheral Nerve Repair: Surgical Applications, State of the Art, and Future Challenges. Crit. Rev. Biomed. Eng. 2011;39:81–124. doi: 10.1615/CritRevBiomedEng.v39.i2.20. - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

