Folic Acid and Vitamin B12 Prevent Deleterious Effects of Rotenone on Object Novelty Recognition Memory and Kynu Expression in an Animal Model of Parkinson's Disease
- PMID: 36553663
- PMCID: PMC9778036
- DOI: 10.3390/genes13122397
Folic Acid and Vitamin B12 Prevent Deleterious Effects of Rotenone on Object Novelty Recognition Memory and Kynu Expression in an Animal Model of Parkinson's Disease
Abstract
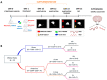
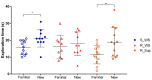
Parkinson's disease (PD) is characterized by a range of motor signs, but cognitive dysfunction is also observed. Supplementation with folic acid and vitamin B12 is expected to prevent cognitive impairment. To test this in PD, we promoted a lesion within the substantia nigra pars compacta of rats using the neurotoxin rotenone. In the sequence, the animals were supplemented with folic acid and vitamin B12 for 14 consecutive days and subjected to the object recognition test. We observed an impairment in object recognition memory after rotenone administration, which was prevented by supplementation (p < 0.01). Supplementation may adjust gene expression through efficient DNA methylation. To verify this, we measured the expression and methylation of the kynureninase gene (Kynu), whose product metabolizes neurotoxic metabolites often accumulated in PD as kynurenine. Supplementation prevented the decrease in Kynu expression induced by rotenone in the substantia nigra (p < 0.05), corroborating the behavioral data. No differences were observed concerning the methylation analysis of two CpG sites in the Kynu promoter. Instead, we suggest that folic acid and vitamin B12 increased global DNA methylation, reduced the expression of Kynu inhibitors, maintained Kynu-dependent pathway homeostasis, and prevented the memory impairment induced by rotenone. Our study raises the possibility of adjuvant therapy for PD with folic acid and vitamin B12.
Keywords: DNA methylation; Kynu; cognitive impairment; gene expression; neurodegenerative diseases; tryptophan pathway.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures



Similar articles
-
Behavioral, neurochemical and histological alterations promoted by bilateral intranigral rotenone administration: a new approach for an old neurotoxin.Neurotox Res. 2012 Apr;21(3):291-301. doi: 10.1007/s12640-011-9278-3. Epub 2011 Sep 28. Neurotox Res. 2012. PMID: 21953489
-
Vortioxetine ameliorates motor and cognitive impairments in the rotenone-induced Parkinson's disease via targeting TLR-2 mediated neuroinflammation.Neuropharmacology. 2022 May 1;208:108977. doi: 10.1016/j.neuropharm.2022.108977. Epub 2022 Jan 26. Neuropharmacology. 2022. PMID: 35092748
-
Protective effect of curcumin against rotenone-induced substantia nigra pars compacta neuronal dysfunction.Metab Brain Dis. 2022 Apr;37(4):1111-1118. doi: 10.1007/s11011-022-00941-6. Epub 2022 Mar 3. Metab Brain Dis. 2022. PMID: 35239141
-
An update on the rotenone models of Parkinson's disease: their ability to reproduce the features of clinical disease and model gene-environment interactions.Neurotoxicology. 2015 Jan;46:101-16. doi: 10.1016/j.neuro.2014.12.002. Epub 2014 Dec 13. Neurotoxicology. 2015. PMID: 25514659 Review.
-
The role of folic acid and Vitamin B12 in genomic stability of human cells.Mutat Res. 2001 Apr 18;475(1-2):57-67. doi: 10.1016/s0027-5107(01)00079-3. Mutat Res. 2001. PMID: 11295154 Review.
Cited by
-
Methionine cycle in C. elegans serotonergic neurons regulates diet-dependent behaviour and longevity through neuron-gut signaling.Nat Commun. 2025 Jun 2;16(1):5118. doi: 10.1038/s41467-025-60475-0. Nat Commun. 2025. PMID: 40456752 Free PMC article.
-
Microbiota, Tryptophan and Aryl Hydrocarbon Receptors as the Target Triad in Parkinson's Disease-A Narrative Review.Int J Mol Sci. 2024 Mar 2;25(5):2915. doi: 10.3390/ijms25052915. Int J Mol Sci. 2024. PMID: 38474162 Free PMC article. Review.
-
Unveiling the Therapeutic Potential of Folate-Dependent One-Carbon Metabolism in Cancer and Neurodegeneration.Int J Mol Sci. 2024 Aug 28;25(17):9339. doi: 10.3390/ijms25179339. Int J Mol Sci. 2024. PMID: 39273288 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

