Dual Role of Fibroblasts Educated by Tumour in Cancer Behavior and Therapeutic Perspectives
- PMID: 36555218
- PMCID: PMC9778751
- DOI: 10.3390/ijms232415576
Dual Role of Fibroblasts Educated by Tumour in Cancer Behavior and Therapeutic Perspectives
Abstract
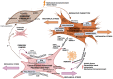
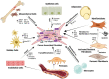
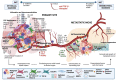
Tumours are complex systems with dynamic interactions between tumour cells, non-tumour cells, and extracellular components that comprise the tumour microenvironment (TME). The majority of TME's cells are cancer-associated fibroblasts (CAFs), which are crucial in extracellular matrix (ECM) construction, tumour metabolism, immunology, adaptive chemoresistance, and tumour cell motility. CAF subtypes have been identified based on the expression of protein markers. CAFs may act as promoters or suppressors in tumour cells depending on a variety of factors, including cancer stage. Indeed, CAFs have been shown to promote tumour growth, survival and spread, and secretome changes, but they can also slow tumourigenesis at an early stage through mechanisms that are still poorly understood. Stromal-cancer interactions are governed by a variety of soluble factors that determine the outcome of the tumourigenic process. Cancer cells release factors that enhance the ability of fibroblasts to secrete multiple tumour-promoting chemokines, acting on malignant cells to promote proliferation, migration, and invasion. This crosstalk between CAFs and tumour cells has given new prominence to the stromal cells, from being considered as mere physical support to becoming key players in the tumour process. Here, we focus on the concept of cancer as a non-healing wound and the relevance of chronic inflammation to tumour initiation. In addition, we review CAFs heterogeneous origins and markers together with the potential therapeutic implications of CAFs "re-education" and/or targeting tumour progression inhibition.
Keywords: cancer cell; cancer-associated fibroblast; inflammation; magnification; metastasis; tumour microenvironment.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

