The Mutual Relationship between Glycosylation and Non-Coding RNAs in Cancer and Other Physio-Pathological Conditions
- PMID: 36555445
- PMCID: PMC9781064
- DOI: 10.3390/ijms232415804
The Mutual Relationship between Glycosylation and Non-Coding RNAs in Cancer and Other Physio-Pathological Conditions
Abstract
Glycosylation, which consists of the enzymatic addition of sugars to proteins and lipids, is one of the most important post-co-synthetic modifications of these molecules, profoundly affecting their activity. Although the presence of carbohydrate chains is crucial for fine-tuning the interactions between cells and molecules, glycosylation is an intrinsically stochastic process regulated by the relative abundance of biosynthetic (glycosyltransferases) and catabolic (glycosidases) enzymes, as well as sugar carriers and other molecules. Non-coding RNAs, which include microRNAs, long non-coding RNAs and circRNAs, establish a complex network of reciprocally interacting molecules whose final goal is the regulation of mRNA expression. Likewise, these interactions are stochastically regulated by ncRNA abundance. Thus, while protein sequence is deterministically dictated by the DNA/RNA/protein axis, protein abundance and activity are regulated by two stochastic processes acting, respectively, before and after the biosynthesis of the protein axis. Consequently, the worlds of glycosylation and ncRNA are closely interconnected and mutually interacting. In this paper, we will extensively review the many faces of the ncRNA-glycosylation interplay in cancer and other physio-pathological conditions.
Keywords: glycosylation; glycosyltransferases; miRNA; non-coding RNAs; sugar antigens.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



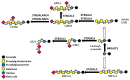

Similar articles
-
A transcriptomic profile of topping responsive non-coding RNAs in tobacco roots (Nicotiana tabacum).BMC Genomics. 2019 Nov 14;20(1):856. doi: 10.1186/s12864-019-6236-6. BMC Genomics. 2019. PMID: 31726968 Free PMC article.
-
Insights into miRNA regulation of the human glycome.Biochem Biophys Res Commun. 2014 Mar 21;445(4):774-9. doi: 10.1016/j.bbrc.2014.01.034. Epub 2014 Jan 23. Biochem Biophys Res Commun. 2014. PMID: 24463102 Free PMC article.
-
Comprehensive analysis of the whole coding and non-coding RNA transcriptome expression profiles and construction of the circRNA-lncRNA co-regulated ceRNA network in laryngeal squamous cell carcinoma.Funct Integr Genomics. 2019 Jan;19(1):109-121. doi: 10.1007/s10142-018-0631-y. Epub 2018 Aug 21. Funct Integr Genomics. 2019. PMID: 30128795
-
The Nefarious Nexus of Noncoding RNAs in Cancer.Int J Mol Sci. 2018 Jul 17;19(7):2072. doi: 10.3390/ijms19072072. Int J Mol Sci. 2018. PMID: 30018188 Free PMC article. Review.
-
Noncoding RNAs: New Players in Cancers.Adv Exp Med Biol. 2016;927:1-47. doi: 10.1007/978-981-10-1498-7_1. Adv Exp Med Biol. 2016. PMID: 27376730 Review.
Cited by
-
Non-Coding RNAs and Innate Immune Responses in Cancer.Biomedicines. 2024 Sep 11;12(9):2072. doi: 10.3390/biomedicines12092072. Biomedicines. 2024. PMID: 39335585 Free PMC article. Review.
-
MAN2B1 in immune system-related diseases, neurodegenerative disorders and cancers: functions beyond α-mannosidosis.Expert Rev Mol Med. 2024 Dec 4;27:e4. doi: 10.1017/erm.2024.34. Expert Rev Mol Med. 2024. PMID: 39628046 Free PMC article. Review.
-
Glycosylation in cancer: mechanisms, diagnostic markers, and therapeutic applications.Mol Cell Biochem. 2025 May 19. doi: 10.1007/s11010-025-05303-1. Online ahead of print. Mol Cell Biochem. 2025. PMID: 40389792 Review.
-
Comprehensive analysis of glycoprotein profiles and their association with cardiovascular disease-related microRNAs in rheumatoid arthritis, metabolic disorders, and controls.Sci Rep. 2024 Nov 2;14(1):26471. doi: 10.1038/s41598-024-77772-1. Sci Rep. 2024. PMID: 39488576 Free PMC article.
-
The Post-Translational Modifications of Human Salivary Peptides and Proteins Evidenced by Top-Down Platforms.Int J Mol Sci. 2023 Aug 14;24(16):12776. doi: 10.3390/ijms241612776. Int J Mol Sci. 2023. PMID: 37628956 Free PMC article. Review.
References
-
- Flynn R.A., Pedram K., Malaker S.A., Batista P.J., Smith B.A.H., Johnson A.G., George B.M., Majzoub K., Villalta P.W., Carette J.E., et al. Small RNAs are modified with N-glycans and displayed on the surface of living cells. Cell. 2021;184:3109–3124. doi: 10.1016/j.cell.2021.04.023. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

