The Challenges of O2 Detection in Biological Fluids: Classical Methods and Translation to Clinical Applications
- PMID: 36555613
- PMCID: PMC9786805
- DOI: 10.3390/ijms232415971
The Challenges of O2 Detection in Biological Fluids: Classical Methods and Translation to Clinical Applications
Abstract
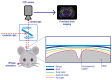
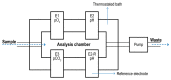
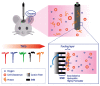
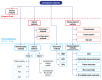
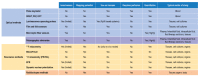
Dissolved oxygen (DO) is deeply involved in preserving the life of cellular tissues and human beings due to its key role in cellular metabolism: its alterations may reflect important pathophysiological conditions. DO levels are measured to identify pathological conditions, explain pathophysiological mechanisms, and monitor the efficacy of therapeutic approaches. This is particularly relevant when the measurements are performed in vivo but also in contexts where a variety of biological and synthetic media are used, such as ex vivo organ perfusion. A reliable measurement of medium oxygenation ensures a high-quality process. It is crucial to provide a high-accuracy, real-time method for DO quantification, which could be robust towards different medium compositions and temperatures. In fact, biological fluids and synthetic clinical fluids represent a challenging environment where DO interacts with various compounds and can change continuously and dynamically, and further precaution is needed to obtain reliable results. This study aims to present and discuss the main oxygen detection and quantification methods, focusing on the technical needs for their translation to clinical practice. Firstly, we resumed all the main methodologies and advancements concerning dissolved oxygen determination. After identifying the main groups of all the available techniques for DO sensing based on their mechanisms and applicability, we focused on transferring the most promising approaches to a clinical in vivo/ex vivo setting.
Keywords: absolute and relative techniques; biological fluids; clinical applications; dissolved oxygen quantification; oxygen sensing; technical innovation in clinics.
Conflict of interest statement
The authors declare no conflict of interest. Valentina Marassi, Pierluigi Reschiglian, Barbara Roda, and Andrea Zattoni are associates of the spinoff company byFlow srl; the company mission includes know-how transfer, development, and application of novel technologies and methodologies for the analysis and characterization of samples of nano-biotechnological interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures


Similar articles
-
Recognising Potential Ambiguities in Measurements of Oxygen in Tissues.Adv Exp Med Biol. 2024;1463:307-314. doi: 10.1007/978-3-031-67458-7_50. Adv Exp Med Biol. 2024. PMID: 39400840
-
Development of a new clinically applicable device for embryo evaluation which measures embryo oxygen consumption.Hum Reprod. 2016 Oct;31(10):2321-30. doi: 10.1093/humrep/dew187. Epub 2016 Sep 8. Hum Reprod. 2016. PMID: 27609982
-
[Myocardial microcirculation in humans--new approaches using MRI].Herz. 2003 Mar;28(2):74-81. doi: 10.1007/s00059-003-2451-6. Herz. 2003. PMID: 12669220 German.
-
From system to organ to cell: oxygenation and perfusion measurement in anesthesia and critical care.J Clin Monit Comput. 2012 Aug;26(4):255-65. doi: 10.1007/s10877-012-9350-4. Epub 2012 Mar 22. J Clin Monit Comput. 2012. PMID: 22437884 Free PMC article. Review.
-
Oxygen demand of perfused heart preparations: how electromechanical function and inadequate oxygenation affect physiology and optical measurements.Exp Physiol. 2015 Jun;100(6):603-16. doi: 10.1113/EP085042. Exp Physiol. 2015. PMID: 25865254 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

