Receptor-Independent Anti-Ferroptotic Activity of TrkB Modulators
- PMID: 36555849
- PMCID: PMC9784883
- DOI: 10.3390/ijms232416205
Receptor-Independent Anti-Ferroptotic Activity of TrkB Modulators
Abstract
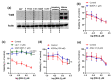
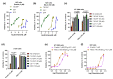
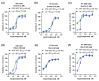
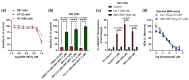
Dysregulated brain-derived neurotrophic factor (BDNF)/tropomyosin receptor kinase B (TrkB) signalling is implicated in several neurodegenerative diseases, including Alzheimer's disease. A failure of neurotrophic support may participate in neurodegenerative mechanisms, such as ferroptosis, which has likewise been implicated in this disease class. The current study investigated whether modulators of TrkB signalling affect ferroptosis. Cell viability, C11 BODIPY, and cell-free oxidation assays were used to observe the impact of TrkB modulators, and an immunoblot assay was used to detect TrkB expression. TrkB modulators such as agonist BDNF, antagonist ANA-12, and inhibitor K252a did not affect RSL3-induced ferroptosis sensitivity in primary cortical neurons expressing detectable TrkB receptors. Several other modulators of the TrkB receptor, including agonist 7,8-DHF, activator phenelzine sulphate, and inhibitor GNF-5837, conferred protection against a range of ferroptosis inducers in several immortalised neuronal and non-neuronal cell lines, such as N27 and HT-1080 cells. We found these immortalised cell lines lack detectable TrkB receptor expression, so the anti-ferroptotic activity of these TrkB modulators was most likely due to their inherent radical-trapping antioxidant properties, which should be considered when interpreting their experimental findings. These modulators or their variants could be potential anti-ferroptotic therapeutics for various diseases.
Keywords: BDNF; TrkB; agonist; antagonist; ferroptosis; inhibitor; protection.
Conflict of interest statement
A.I.B. owns equity in Alterity Ltd., Cogstate Ltd., and a profit share in Collaborative Medicinal Development LLC.
Figures

References
-
- Jerónimo-Santos A., Vaz S.H., Parreira S., Rapaz-Lérias S., Caetano A.P., Buée-Scherrer V., Castrén E., Valente C.A., Blum D., Sebastião A.M., et al. Dysregulation of trkb receptors and bdnf function by amyloid-β peptide is mediated by calpain. Cereb. Cortex. 2014;25:3107–3121. doi: 10.1093/cercor/bhu105. - DOI - PubMed
-
- Xiang J., Wang Z.H., Ahn E.H., Liu X., Yu S.P., Manfredsson F.P., Sandoval I.M., Ju G., Wu S., Ye K. Delta-secretase-cleaved tau antagonizes trkb neurotrophic signalings, mediating Alzheimer’s disease pathologies. Proc. Natl. Acad. Sci. USA. 2019;116:9094–9102. doi: 10.1073/pnas.1901348116. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

