Naringin Attenuates the Diabetic Neuropathy in STZ-Induced Type 2 Diabetic Wistar Rats
- PMID: 36556476
- PMCID: PMC9782177
- DOI: 10.3390/life12122111
Naringin Attenuates the Diabetic Neuropathy in STZ-Induced Type 2 Diabetic Wistar Rats
Abstract
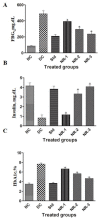
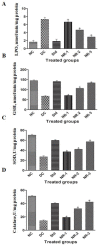
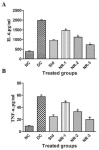
The application of traditional medicines for the treatment of diseases, including diabetic neuropathy (DN), has received great attention. The aim of this study was to investigate the ameliorative potential of naringin, a flavanone, to treat streptozotocin-induced DN in rat models. After the successful induction of diabetes, DN complications were measured by various behavioral tests after 4 weeks of post-induction of diabetes with or without treatment with naringin. Serum biochemical assays such as fasting blood glucose, HbA1c%, insulin, lipid profile, and oxidative stress parameters were determined. Proinflammatory cytokines such as TNF-α and IL-6, and neuron-specific markers such as BDNF and NGF, were also assessed. In addition, pancreatic and brain tissues were subjected to histopathology to analyze structural alterations. The diabetic rats exhibited increased paw withdrawal frequencies for the acetone drop test and decreased frequencies for the plantar test, hot plate test, and tail flick test. The diabetic rats also showed an altered level of proinflammatory cytokines and oxidative stress parameters, as well as altered levels of proinflammatory cytokines and oxidative stress parameters. Naringin treatment significantly improved these parameters and helped in restoring the normal architecture of the brain and pancreatic tissues. The findings show that naringin's neuroprotective properties may be linked to its ability to suppress the overactivation of inflammatory molecules and mediators of oxidative stress.
Keywords: diabetes mellitus; diabetic neuropathy; histopathology; naringin; neuronal specific markers; proinflammatory cytokines; streptozotocin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Neuroprotective effects of Gymnema sylvestre on streptozotocin-induced diabetic neuropathy in rats.Exp Ther Med. 2015 May;9(5):1670-1678. doi: 10.3892/etm.2015.2305. Epub 2015 Feb 20. Exp Ther Med. 2015. PMID: 26136876 Free PMC article.
-
Naringenin neutralises oxidative stress and nerve growth factor discrepancy in experimental diabetic neuropathy.Neurol Res. 2015 Oct;37(10):924-33. doi: 10.1179/1743132815Y.0000000079. Epub 2015 Jul 17. Neurol Res. 2015. PMID: 26187552
-
Hesperidin and naringin attenuate hyperglycemia-mediated oxidative stress and proinflammatory cytokine production in high fat fed/streptozotocin-induced type 2 diabetic rats.J Diabetes Complications. 2012 Nov-Dec;26(6):483-90. doi: 10.1016/j.jdiacomp.2012.06.001. Epub 2012 Jul 17. J Diabetes Complications. 2012. PMID: 22809898
-
The potential of esculin in ameliorating Type-2 diabetes mellitus induced neuropathy in Wistar rats and probing its inhibitory mechanism of insulin aggregation.Int J Biol Macromol. 2023 Jul 1;242(Pt 1):124760. doi: 10.1016/j.ijbiomac.2023.124760. Epub 2023 May 7. Int J Biol Macromol. 2023. PMID: 37156314
-
Naringin: Nanotechnological Strategies for Potential Pharmaceutical Applications.Pharmaceutics. 2023 Mar 7;15(3):863. doi: 10.3390/pharmaceutics15030863. Pharmaceutics. 2023. PMID: 36986723 Free PMC article. Review.
Cited by
-
Diabetic Neuropathy: An Overview of Molecular Pathways and Protective Mechanisms of Phytobioactives.Endocr Metab Immune Disord Drug Targets. 2024;24(7):758-776. doi: 10.2174/0118715303266444231008143430. Endocr Metab Immune Disord Drug Targets. 2024. PMID: 37867264 Review.
-
Alpha-lipoic acid modulates the diabetes mellitus-mediated neuropathic pain via inhibition of the TRPV1 channel, apoptosis, and oxidative stress in rats.J Bioenerg Biomembr. 2023 Jun;55(3):179-193. doi: 10.1007/s10863-023-09971-w. Epub 2023 Jun 26. J Bioenerg Biomembr. 2023. PMID: 37357235
-
Mitigative effect of sodium alginate on streptozotocin (STZ)-induced diabetic neuropathy through regulation of redox status and miR-146a in the rat sciatic nerve.PeerJ. 2025 Mar 24;13:e19046. doi: 10.7717/peerj.19046. eCollection 2025. PeerJ. 2025. PMID: 40151454 Free PMC article.
-
Flavonoids regulating NLRP3 inflammasome: a promising approach in alleviating diabetic peripheral neuropathy.Inflammopharmacology. 2025 May;33(5):2231-2262. doi: 10.1007/s10787-025-01729-7. Epub 2025 Apr 9. Inflammopharmacology. 2025. PMID: 40205269 Free PMC article. Review.
-
Attenuation of Streptozotocin-Induced Diabetic Neuropathic Allodynia by Flavone Derivative Through Modulation of GABA-ergic Mechanisms and Endogenous Biomarkers.Neurochem Res. 2024 Apr;49(4):980-997. doi: 10.1007/s11064-023-04078-5. Epub 2024 Jan 3. Neurochem Res. 2024. PMID: 38170385
References
-
- Edwards J.R., Peterson K.D., Mu Y., Banerjee S., Allen-Bridson K., Morrell G., Dudeck M.A., Pollock D.A., Horan T.C. National Healthcare Safety Network (NHSN) report: Data summary for 2006 through 2008, issued December 2009. Am. J. Infect. Control. 2009;37:783–805. doi: 10.1016/j.ajic.2009.10.001. - DOI - PubMed
LinkOut - more resources
Full Text Sources

