Metabolomics and Lipidomics Signatures of Insulin Resistance and Abdominal Fat Depots in People Living with Obesity
- PMID: 36557310
- PMCID: PMC9781703
- DOI: 10.3390/metabo12121272
Metabolomics and Lipidomics Signatures of Insulin Resistance and Abdominal Fat Depots in People Living with Obesity
Abstract
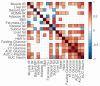
The liver, skeletal muscle, and adipose tissue are major insulin target tissues and key players in glucose homeostasis. We and others have described diverse insulin resistance (IR) phenotypes in people at risk of developing type 2 diabetes. It is postulated that identifying the IR phenotype in a patient may guide the treatment or the prevention strategy for better health outcomes in populations at risk. Here, we performed plasma metabolomics and lipidomics in a cohort of men and women living with obesity not complicated by diabetes (mean [SD] BMI 36.0 [4.5] kg/m2, n = 62) to identify plasma signatures of metabolites and lipids that align with phenotypes of IR (muscle, liver, or adipose tissue) and abdominal fat depots. We used 2-step hyperinsulinemic-euglycemic clamp with deuterated glucose, oral glucose tolerance test, dual-energy X-ray absorptiometry and abdominal magnetic resonance imaging to assess muscle-, liver- and adipose tissue- IR, beta cell function, body composition, abdominal fat distribution and liver fat, respectively. Spearman’s rank correlation analyses that passed the Benjamini−Hochberg statistical correction revealed that cytidine, gamma-aminobutyric acid, anandamide, and citrate corresponded uniquely with muscle IR, tryptophan, cAMP and phosphocholine corresponded uniquely with liver IR and phenylpyruvate and hydroxy-isocaproic acid corresponded uniquely with adipose tissue IR (p < 7.2 × 10−4). Plasma cholesteryl sulfate (p = 0.00029) and guanidinoacetic acid (p = 0.0001) differentiated between visceral and subcutaneous adiposity, while homogentisate correlated uniquely with liver fat (p = 0.00035). Our findings may help identify diverse insulin resistance and adiposity phenotypes and enable targeted treatments in people living with obesity.
Keywords: abdominal fat deposition; liver fat; obesity phenotypes; plasma lipidomics; plasma metabolomics; tissue insulin resistance.
Conflict of interest statement
The authors declared no conflict of interest.
Figures


References
-
- Groop L.C., Bonadonna R.C., DelPrato S., Ratheiser K., Zyck K., Ferrannini E., DeFronzo R.A. Glucose and free fatty acid metabolism in non-insulin-dependent diabetes mellitus. Evidence for multiple sites of insulin resistance. J. Clin. Investig. 1989;84:205–213. doi: 10.1172/JCI114142. - DOI - PMC - PubMed
-
- Blanco-Rojo R., Alcala-Diaz J.F., Wopereis S., Perez-Martinez P., Quintana-Navarro G.M., Marin C., Ordovas J.M., van Ommen B., Perez-Jimenez F., Delgado-Lista J., et al. The insulin resistance phenotype (muscle or liver) interacts with the type of diet to determine changes in disposition index after 2 years of intervention: The CORDIOPREV-DIAB randomised clinical trial. Diabetologia. 2016;59:67–76. doi: 10.1007/s00125-015-3776-4. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

