Ubiquitin-Conjugating Enzymes Ubc1 and Ubc4 Mediate the Turnover of Hap4, a Master Regulator of Mitochondrial Biogenesis in Saccharomyces cerevisiae
- PMID: 36557625
- PMCID: PMC9787919
- DOI: 10.3390/microorganisms10122370
Ubiquitin-Conjugating Enzymes Ubc1 and Ubc4 Mediate the Turnover of Hap4, a Master Regulator of Mitochondrial Biogenesis in Saccharomyces cerevisiae
Abstract
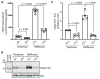
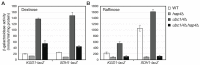
Mitochondrial biogenesis is tightly regulated in response to extracellular and intracellular signals, thereby adapting yeast cells to changes in their environment. The Hap2/3/4/5 complex is a master transcriptional regulator of mitochondrial biogenesis in yeast. Hap4 is the regulatory subunit of the complex and exhibits increased expression when the Hap2/3/4/5 complex is activated. In cells grown under glucose derepression conditions, both the HAP4 transcript level and Hap4 protein level are increased. As part of an inter-organellar signaling mechanism coordinating gene expression between the mitochondrial and nuclear genomes, the activity of the Hap2/3/4/5 complex is reduced in respiratory-deficient cells, such as ρ0 cells lacking mitochondrial DNA, as a result of reduced Hap4 protein levels. However, the underlying mechanism is unclear. Here, we show that reduced HAP4 expression in ρ0 cells is mediated through both transcriptional and post-transcriptional mechanisms. We show that loss of mitochondrial DNA increases the turnover of Hap4, which requires the 26S proteasome and ubiquitin-conjugating enzymes Ubc1 and Ubc4. Stabilization of Hap4 in the ubc1 ubc4 double mutant leads to increased expression of Hap2/3/4/5-target genes. Our results indicate that mitochondrial biogenesis in yeast is regulated by the functional state of mitochondria partly through ubiquitin/proteasome-dependent turnover of Hap4.
Keywords: Hap4; Saccharomyces cerevisiae; Ubc1; Ubc4; mitochondrial dysfunction; proteasome; protein turnover; transcription factor; ubiquitin-conjugating enzyme (E2 enzyme).
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
An E2 accessory domain increases affinity for the anaphase-promoting complex and ensures E2 competition.J Biol Chem. 2015 Oct 2;290(40):24614-25. doi: 10.1074/jbc.M115.678193. Epub 2015 Aug 25. J Biol Chem. 2015. PMID: 26306044 Free PMC article.
-
Positive regulation of the LPD1 gene of Saccharomyces cerevisiae by the HAP2/HAP3/HAP4 activation system.Mol Gen Genet. 1992 Jan;231(2):296-303. doi: 10.1007/BF00279803. Mol Gen Genet. 1992. PMID: 1310523
-
UBC1 encodes a novel member of an essential subfamily of yeast ubiquitin-conjugating enzymes involved in protein degradation.EMBO J. 1990 Dec;9(13):4535-41. doi: 10.1002/j.1460-2075.1990.tb07905.x. EMBO J. 1990. PMID: 2265617 Free PMC article.
-
Yeast ubiquitin-conjugating enzymes involved in selective protein degradation are essential for cell viability.Acta Biol Hung. 1991;42(1-3):27-37. Acta Biol Hung. 1991. PMID: 1844315 Review.
-
Genetic analysis of ubiquitin-dependent protein degradation.Experientia. 1992 Feb 15;48(2):172-8. doi: 10.1007/BF01923510. Experientia. 1992. PMID: 1740189 Review.
Cited by
-
Inactivation of HAP4 Accelerates RTG-Dependent Osmoadaptation in Saccharomyces cerevisiae.Int J Mol Sci. 2023 Mar 10;24(6):5320. doi: 10.3390/ijms24065320. Int J Mol Sci. 2023. PMID: 36982394 Free PMC article.
-
GSM1 Requires Hap4 for Expression and Plays a Role in Gluconeogenesis and Utilization of Nonfermentable Carbon Sources.Genes (Basel). 2024 Aug 27;15(9):1128. doi: 10.3390/genes15091128. Genes (Basel). 2024. PMID: 39336719 Free PMC article.
-
Lifespan regulation by targeting heme signaling in yeast.Geroscience. 2024 Oct;46(5):5235-5245. doi: 10.1007/s11357-024-01218-9. Epub 2024 May 29. Geroscience. 2024. PMID: 38809391 Free PMC article.
-
Lifespan regulation by targeting heme signaling in yeast.bioRxiv [Preprint]. 2024 Jan 21:2024.01.20.576446. doi: 10.1101/2024.01.20.576446. bioRxiv. 2024. Update in: Geroscience. 2024 Oct;46(5):5235-5245. doi: 10.1007/s11357-024-01218-9. PMID: 38293148 Free PMC article. Updated. Preprint.
References
-
- Woo D.K., Phang T.L., Trawick J.D., Poyton R.O. Multiple pathways of mitochondrial-nuclear communication in yeast: Intergenomic signaling involves ABF1 and affects a different set of genes than retrograde regulation. Biochim. Biophys. Acta. 2009;1789:135–145. doi: 10.1016/j.bbagrm.2008.09.008. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

