Coumarin-Induced Hepatotoxicity: A Narrative Review
- PMID: 36558195
- PMCID: PMC9783661
- DOI: 10.3390/molecules27249063
Coumarin-Induced Hepatotoxicity: A Narrative Review
Abstract
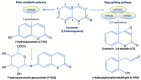
Coumarin is an effective treatment for primary lymphoedema, as well as lymphoedema related to breast cancer radiotherapy or surgery. However, its clinical use is limited in several countries due to the possible occurrence of hepatotoxicity, mainly in the form of mild to moderate transaminase elevation. It is worth noting that only a few cases of severe hepatotoxicity have been described in the literature, with no reported cases of liver failure. Data available on coumarin absorption, distribution, metabolism, and excretion have been reviewed, focusing on hepatotoxicity studies carried out in vitro and in vivo. Finally, safety and tolerability data from clinical trials have been thoroughly discussed. Based on these data, coumarin-induced hepatotoxicity is restricted to a small subset of patients, probably due to the activation in these individuals of alternative metabolic pathways involving specific CYP450s isoforms. The aim of this work is to stimulate research to clearly identify patients at risk of developing hepatotoxicity following coumarin treatment. Early identification of this subset of patients could open the possibility of more safely exploiting the therapeutical properties of coumarin, allowing patients suffering from lymphoedema to benefit from the anti-oedematous activity of the treatment.
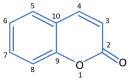
Keywords: 1,2-benzopyrone; Melilotus officinalis; coumarin; hepatotoxicity; narrative review; primary lymphoedema; secondary lymphoedema.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
-
- Bell R.G. Metabolism of Vitamin K and Prothrombin Synthesis: Anticoagulants and the Vitamin K--Epoxide Cycle. Fed. Proc. 1978;37:2599–2604. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

