Impact of Dietary Arachidonic Acid on Gut Microbiota Composition and Gut-Brain Axis in Male BALB/C Mice
- PMID: 36558497
- PMCID: PMC9786182
- DOI: 10.3390/nu14245338
Impact of Dietary Arachidonic Acid on Gut Microbiota Composition and Gut-Brain Axis in Male BALB/C Mice
Abstract
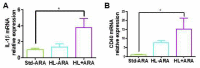
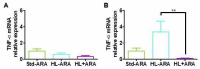
Although arachidonic acid (ARA) is the precursor of the majority of eicosanoids, its influence as a food component on health is not well known. Therefore, we investigated its impact on the gut microbiota and gut-brain axis. Groups of male BALB/c mice were fed either a standard diet containing 5% lipids (Std-ARA) or 15%-lipid diets without ARA (HL-ARA) or with 1% ARA (HL + ARA) for 9 weeks. Fatty acid profiles of all three diets were the same. The HL-ARA diet favored the growth of Bifidobacterium pseudolongum contrary to the HL + ARA diet that favored the pro-inflammatory Escherichia-Shigella genus in fecal microbiota. Dietary ARA intake induced 4- and 15-fold colic overexpression of the pro-inflammatory markers IL-1β and CD40, respectively, without affecting those of TNFα and adiponectin. In the brain, dietary ARA intake led to moderate overexpression of GFAP in the hippocampus and cortex. Both the hyperlipidic diets reduced IL-6 and IL-12 in the brain. For the first time, it was shown that dietary ARA altered the gut microbiota, led to low-grade colic inflammation, and induced astrogliosis in the brain. Further work is necessary to determine the involved mechanisms.
Keywords: arachidonic acid; gut microbiota; gut–brain axis; hyperlipidic diet; inflammation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Effects of dietary arachidonic acid in European sea bass (Dicentrarchus labrax) distal intestine lipid classes and gut health.Fish Physiol Biochem. 2020 Apr;46(2):681-697. doi: 10.1007/s10695-019-00744-0. Epub 2019 Dec 16. Fish Physiol Biochem. 2020. PMID: 31845079
-
Arachidonic acid sex-dependently affects obesity through linking gut microbiota-driven inflammation to hypothalamus-adipose-liver axis.Biochim Biophys Acta Mol Basis Dis. 2017 Nov;1863(11):2715-2726. doi: 10.1016/j.bbadis.2017.07.003. Epub 2017 Jul 12. Biochim Biophys Acta Mol Basis Dis. 2017. PMID: 28711599
-
2'-fucosyllactose Supplementation Improves Gut-Brain Signaling and Diet-Induced Obese Phenotype and Changes the Gut Microbiota in High Fat-Fed Mice.Nutrients. 2020 Apr 5;12(4):1003. doi: 10.3390/nu12041003. Nutrients. 2020. PMID: 32260563 Free PMC article.
-
Gut microbes participate in food preference alterations during obesity.Gut Microbes. 2021 Jan-Dec;13(1):1959242. doi: 10.1080/19490976.2021.1959242. Gut Microbes. 2021. PMID: 34424831 Free PMC article. Review.
-
Dietary Influences on the Microbiota-Gut-Brain Axis.Int J Mol Sci. 2021 Mar 28;22(7):3502. doi: 10.3390/ijms22073502. Int J Mol Sci. 2021. PMID: 33800707 Free PMC article. Review.
Cited by
-
Can essential fatty acids (EFAs) prevent and ameliorate post-COVID-19 long haul manifestations?Lipids Health Dis. 2024 Apr 19;23(1):112. doi: 10.1186/s12944-024-02090-4. Lipids Health Dis. 2024. PMID: 38641607 Free PMC article. Review.
-
Trans, Trans-Farnesol Enhances the Anti-Bacterial and Anti-Biofilm Effect of Arachidonic Acid on the Cariogenic Bacteria Streptococcus mutans and Streptococcus sobrinus.Int J Mol Sci. 2024 Nov 1;25(21):11770. doi: 10.3390/ijms252111770. Int J Mol Sci. 2024. PMID: 39519322 Free PMC article.
-
Persistent immune dysregulation and metabolic alterations following SARS-CoV-2 infection.medRxiv [Preprint]. 2025 Apr 17:2025.04.16.25325949. doi: 10.1101/2025.04.16.25325949. medRxiv. 2025. PMID: 40321289 Free PMC article. Preprint.
References
-
- Calder P.C., Campoy C., Eilander A., Fleith M., Forsyth S., Larsson P.O., Schelkle B., Lohner S., Szommer A., van de Heijning B.J.M., et al. A systematic review of the effects of increasing arachidonic acid intake on PUFA status, metabolism and health-related outcomes in humans. Br. J. Nutr. 2019;121:1201–1214. doi: 10.1017/S0007114519000692. - DOI - PubMed
-
- Mariamenatu A.H., Abdu E.M. Overconsumption of Omega-6 Polyunsaturated Fatty Acids (PUFAs) versus Deficiency of Omega-3 PUFAs in Modern-Day Diets: The Disturbing Factor for Their “Balanced Antagonistic Metabolic Function” in the Human Body. J. Lipids. 2021;2021:8848161. doi: 10.1155/2021/8848161. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

