Co-Infection of the Epstein-Barr Virus and the Kaposi Sarcoma-Associated Herpesvirus
- PMID: 36560713
- PMCID: PMC9782805
- DOI: 10.3390/v14122709
Co-Infection of the Epstein-Barr Virus and the Kaposi Sarcoma-Associated Herpesvirus
Abstract
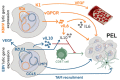
The two human tumor viruses, Epstein-Barr virus (EBV) and Kaposi sarcoma-associated herpesvirus (KSHV), have been mostly studied in isolation. Recent studies suggest that co-infection with both viruses as observed in one of their associated malignancies, namely primary effusion lymphoma (PEL), might also be required for KSHV persistence. In this review, we discuss how EBV and KSHV might support each other for persistence and lymphomagenesis. Moreover, we summarize what is known about their innate and adaptive immune control which both seem to be required to ensure asymptomatic persistent co-infection with these two human tumor viruses. A better understanding of this immune control might allow us to prepare for vaccination against EBV and KSHV in the future.
Keywords: B cell lymphomas; Kaposi sarcoma; T cells; cytotoxic lymphocytes; humanized mice; latent and lytic infection; natural killer cells.
Conflict of interest statement
The author declares no conflict of interest.
Figures

References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

