Airway Inflammation Before and After Bronchial Thermoplasty in Severe Asthma
- PMID: 36560975
- PMCID: PMC9767029
- DOI: 10.2147/JAA.S383418
Airway Inflammation Before and After Bronchial Thermoplasty in Severe Asthma
Abstract
Background: Bronchial thermoplasty (BT) is a bronchoscopic treatment for severe asthma, of which the working mechanism and responder profile are partly unknown. The aim of this study is to analyse whether BT alters airway inflammation by epithelial gene expression, inflammatory cell counts and cytokines, and whether this relates to treatment response.
Methods: In this clinical trial, 28 severe asthma patients underwent bronchoscopy before and after treatment to obtain bronchial brushes and bronchoalveolar lavage fluid (BALF) from treated and untreated airways. RNA was extracted from bronchial brushes for transcriptome analysis, and BALF cells and cytokines were analysed. Asthma quality of life questionnaires were used to distinguish responders from non-responders. We compared results before and after treatment, between treated and untreated airways, and between responders and non-responders.
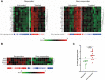
Results: Gene expression of airway epithelium related to airway inflammation gene set was significantly downregulated in treated airways compared to untreated airways, although this did not differ for patients before and after treatment. No differences were observed in cell counts and cytokines, neither from the untreated compared to treated airways, nor before and after treatment. At baseline, compared to non-responders, the expression of genes related to glycolysis in bronchial epithelium was downregulated and both BALF and blood eosinophil counts were higher in responders.
Conclusion: Local differences in gene sets pertaining to epithelial inflammatory status were identified between treated and untreated airways after treatment, not resulting in changes in differential cell counts and cytokine analyses in BALF. Secondly, baseline epithelial glycolysis genes and eosinophil counts in BALF and blood were different between responders and non-responders. The observations from this study demonstrate the potential impact of BT on epithelial gene expression related to airway inflammation while also identifying a possible responder profile.
Keywords: airway inflammation; bronchial thermoplasty; bronchoalveolar lavage fluid cells; cytokines; epithelial transcriptome.
© 2022 Wijsman et al.
Conflict of interest statement
René Lutter and Peter I Bonta are co-last authors for this study. Prof. Dr. Elisabeth H Bel reports grants from Teva, GSK, AstraZeneca, Sanofi, Chiesi, and Teva, outside the submitted work. Dr Peter I Bonta reports grants from Boston Scientific, Stichting Asthma Bestrijding, and Dutch Lung Foundation for Health Research and Development, during the conduct of the study. The authors report no other conflicts of interest in this work.
Figures



References
-
- Global Initiative for Asthma. Global strategy for asthma management and prevention; 2021. Available from: www.ginasthma.org. Accessed December 1, 2022.
-
- Agache I, Beltran J, Akdis C, et al. Efficacy and safety of treatment with biologicals (benralizumab, dupilumab, mepolizumab, omalizumab and reslizumab) for severe eosinophilic asthma. A systematic review for the EAACI Guidelines - recommendations on the use of biologicals in severe asthma. Allergy. 2020;75(5):1023–1042. doi: 10.1111/all.14221 - DOI - PubMed
LinkOut - more resources
Full Text Sources

