Modulation of sirtuins during monolayer chondrocyte culture influences cartilage regeneration upon transfer to a 3D culture environment
- PMID: 36561039
- PMCID: PMC9763269
- DOI: 10.3389/fbioe.2022.971932
Modulation of sirtuins during monolayer chondrocyte culture influences cartilage regeneration upon transfer to a 3D culture environment
Abstract
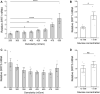
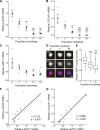
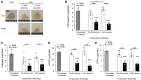
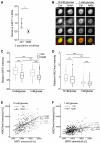
This study examined the role of sirtuins in the regenerative potential of articular chondrocytes. Sirtuins (SIRT1-7) play a key role in regulating cartilage homeostasis. By inhibiting pro-inflammatory pathways responsible for cartilage degradation and promoting the expression of key matrix components, sirtuins have the potential to drive a favourable balance between anabolic and catabolic processes critical to regenerative medicine. When subjected to osmolarity and glucose concentrations representative of the in vivo niche, freshly isolated bovine chondrocytes exhibited increases in SIRT1 but not SIRT3 gene expression. Replicating methods adopted for the in vitro monolayer expansion of chondrocytes for cartilage regenerative therapies, we found that SIRT1 gene expression declined during expansion. Manipulation of sirtuin activity during in vitro expansion by supplementation with the SIRT1-specific activator SRT1720, nicotinamide mononucleotide, or the pan-sirtuin inhibitor nicotinamide, significantly influenced cartilage regeneration in subsequent 3D culture. Tissue mass, cellularity and extracellular matrix content were reduced in response to sirtuin inhibition during expansion, whilst sirtuin activation enhanced these measures of cartilage tissue regeneration. Modulation of sirtuin activity during monolayer expansion influenced H3K27me3, a heterochromatin mark with an important role in development and differentiation. Unexpectedly, treatment of primary chondrocytes with sirtuin activators in 3D culture reduced their matrix synthesis. Thus, modulating sirtuin activity during the in vitro monolayer expansion phase may represent a distinct opportunity to enhance the outcome of cartilage regenerative medicine techniques.
Keywords: cartilage tissue engineering; chondrocyte; glucose restriction; nicotinamide adenine dinucleotide; pellet culture; sirtuin.
Copyright © 2022 Heywood, Thorpe, Jeropoulos, Caton and Lee.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
LinkOut - more resources
Full Text Sources

