Uncovering water effects in protein-ligand recognition: importance in the second hydration shell and binding kinetics
- PMID: 36562309
- PMCID: PMC9970846
- DOI: 10.1039/d2cp04584b
Uncovering water effects in protein-ligand recognition: importance in the second hydration shell and binding kinetics
Abstract
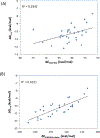
Developing a ligand with high affinity for a specific protein target is essential for drug design, and water molecules are well known to play a key role in protein-drug recognition. However, predicting the role of particularly ordered water molecules in drug binding remains challenging. Furthermore, hydration free energy contributed from the water network, including the second shell of water molecules, is far from being well studied. In this research we focused on these aspects to accurately and efficiently evaluate water effects in protein-ligand binding affinity. We developed a new strategy using a free-energy calculation method, VM2. We successfully predicted the stable ordered water molecules in a number of protein systems: PDE 10a, HSP90, tryptophan synthase (TRPS), CDK2 and Factor Xa. In some of these, the second shell of water molecules appeared to be critical in protein-ligand binding. We also applied the strategy to largely improve binding free energy calculation using the MM/PBSA method. When applying MM/PBSA alone for two systems, CDK2 and Factor Xa, the computed binding free energy resulted in poor to moderate R2 values with experimental data. However, including water free energy correction greatly improved the free energy calculation. Furthermore, our work helped to explain how xk263 is a 1000 times faster binder to HIVp than ritonavir, a potentially useful tool for investigating binding kinetics. Our studies reveal the importance of fully considering water effects in therapeutic developments in pharmaceutical and biotechnology industries and for fundamental research in protein-ligand recognition.
Conflict of interest statement
CONFLICTS OF INTEREST
There are no conflicts to declare.
Figures






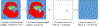

Similar articles
-
Thermodynamic Characterization of Hydration Sites from Integral Equation-Derived Free Energy Densities: Application to Protein Binding Sites and Ligand Series.J Chem Inf Model. 2017 Jul 24;57(7):1652-1666. doi: 10.1021/acs.jcim.6b00765. Epub 2017 Jun 16. J Chem Inf Model. 2017. PMID: 28565907
-
Ritonavir and xk263 Binding-Unbinding with HIV-1 Protease: Pathways, Energy and Comparison.Life (Basel). 2022 Jan 13;12(1):116. doi: 10.3390/life12010116. Life (Basel). 2022. PMID: 35054509 Free PMC article.
-
Molecular recognition in a diverse set of protein-ligand interactions studied with molecular dynamics simulations and end-point free energy calculations.J Chem Inf Model. 2013 Oct 28;53(10):2659-70. doi: 10.1021/ci400312v. Epub 2013 Oct 8. J Chem Inf Model. 2013. PMID: 24032517 Free PMC article.
-
Molecular modeling of hydration in drug design.Curr Opin Drug Discov Devel. 2007 May;10(3):275-80. Curr Opin Drug Discov Devel. 2007. PMID: 17554853 Review.
-
Calculating Water Thermodynamics in the Binding Site of Proteins - Applications of WaterMap to Drug Discovery.Curr Top Med Chem. 2017;17(23):2586-2598. doi: 10.2174/1568026617666170414141452. Curr Top Med Chem. 2017. PMID: 28413953 Review.
Cited by
-
Reliability of AlphaFold2 Models in Virtual Drug Screening: A Focus on Selected Class A GPCRs.Int J Mol Sci. 2024 Sep 21;25(18):10139. doi: 10.3390/ijms251810139. Int J Mol Sci. 2024. PMID: 39337622 Free PMC article.
-
Theoretical Description of Changes in Conformation and Symmetry of Supramolecular Systems During the Reception of a Molecular Signal.Int J Mol Sci. 2025 Jul 3;26(13):6411. doi: 10.3390/ijms26136411. Int J Mol Sci. 2025. PMID: 40650191 Free PMC article.
-
W254 in furin functions as a molecular gate promoting anti-viral drug binding: Elucidation of putative drug tunneling and docking by non-equilibrium molecular dynamics.Comput Struct Biotechnol J. 2023 Sep 9;21:4589-4612. doi: 10.1016/j.csbj.2023.09.003. eCollection 2023. Comput Struct Biotechnol J. 2023. PMID: 37817778 Free PMC article.
-
Enthalpic Classification of Water Molecules in Target-Ligand Binding.J Chem Inf Model. 2024 Aug 26;64(16):6583-6595. doi: 10.1021/acs.jcim.4c00794. Epub 2024 Aug 12. J Chem Inf Model. 2024. PMID: 39135312 Free PMC article.
-
The role of water in mediating DNA structures with epigenetic modifications, higher-order conformations and drug-DNA interactions.RSC Chem Biol. 2025 Mar 14;6(5):699-720. doi: 10.1039/d4cb00308j. eCollection 2025 May 8. RSC Chem Biol. 2025. PMID: 40171245 Free PMC article. Review.
References
-
- Hayashi T Water at Interfaces: Its Behavior and Roles in Interfacial Phenomena. Chemistry Letters, 2021, 50(6): 1173–1180.
-
- Wong S; Lightstone FC Accounting for water molecules in drug design. Expert Opinion on Drug Discovery 2011, 6, 65–74. - PubMed

