PP4 inhibition sensitizes ovarian cancer to NK cell-mediated cytotoxicity via STAT1 activation and inflammatory signaling
- PMID: 36564125
- PMCID: PMC9791393
- DOI: 10.1136/jitc-2022-005026
PP4 inhibition sensitizes ovarian cancer to NK cell-mediated cytotoxicity via STAT1 activation and inflammatory signaling
Erratum in
-
Correction: PP4 inhibition sensitizes ovarian cancer to NK cell-mediated cytotoxicity via STAT1 activation and inflammatory signaling.J Immunother Cancer. 2024 Nov 20;12(11):e005026corr1. doi: 10.1136/jitc-2022-005026corr1. J Immunother Cancer. 2024. PMID: 39572162 Free PMC article. No abstract available.
Abstract
Background: Increased infiltration of T cells into ovarian tumors has been repeatedly shown to be predictive of enhanced patient survival. However, despite the evidence of an active immune response in ovarian cancer (OC), the frequency of responses to immune checkpoint blockade (ICB) therapy in OC is much lower than other cancer types. Recent studies have highlighted that deficiencies in the DNA damage response (DDR) can drive increased genomic instability and tumor immunogenicity, which leads to enhanced responses to ICB. Protein phosphatase 4 (PP4) is a critical regulator of the DDR; however, its potential role in antitumor immunity is currently unknown.
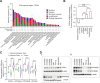
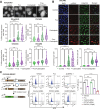
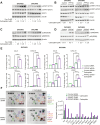
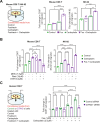
Results: Our results show that the PP4 inhibitor, fostriecin, combined with carboplatin leads to increased carboplatin sensitivity, DNA damage, and micronuclei formation. Using multiple OC cell lines, we show that PP4 inhibition or PPP4C knockdown combined with carboplatin triggers inflammatory signaling via Nuclear factor kappa B (NF-κB) and signal transducer and activator of transcription 1 (STAT1) activation. This resulted in increased expression of the pro-inflammatory cytokines and chemokines: CCL5, CXCL10, and IL-6. In addition, IFNB1 expression was increased suggesting activation of the type I interferon response. Conditioned media from OC cells treated with the combination of PP4 inhibitor and carboplatin significantly increased migration of both CD8 T cell and natural killer (NK) cells over carboplatin treatment alone. Knockdown of stimulator of interferon genes (STING) in OC cells significantly abrogated the increase in CD8 T-cell migration induced by PP4 inhibition. Co-culture of NK-92 cells and OC cells with PPP4C or PPP4R3B knockdown resulted in strong induction of NK cell interferon-γ, increased degranulation, and increased NK cell-mediated cytotoxicity against OC cells. Stable knockdown of PP4C in a syngeneic, immunocompetent mouse model of OC resulted in significantly reduced tumor growth in vivo. Tumors with PP4C knockdown had increased infiltration of NK cells, NK T cells, and CD4+ T cells. Addition of low dose carboplatin treatment led to increased CD8+ T-cell infiltration in PP4C knockdown tumors as compared with the untreated PP4C knockdown tumors.
Conclusions: Our work has identified a role for PP4 inhibition in promoting inflammatory signaling and enhanced immune cell effector function. These findings support the further investigation of PP4 inhibitors to enhance chemo-immunotherapy for OC treatment.
Keywords: Genital Neoplasms, Female; Genome Instability; Immunity, Innate; Inflammation Mediators.
© Author(s) (or their employer(s)) 2022. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: No, there are no competing interests.
Figures




References
-
- Allemani C, Matsuda T, Di Carlo V, et al. Global surveillance of trends in cancer survival 2000-14 (CONCORD-3): analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet. 2018;391:1023–75. doi: 10.1016/S0140-6736(17)33326-3. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
