Improvements in Walking Distance during Nusinersen Treatment - A Prospective 3-year SMArtCARE Registry Study
- PMID: 36565133
- PMCID: PMC9881023
- DOI: 10.3233/JND-221600
Improvements in Walking Distance during Nusinersen Treatment - A Prospective 3-year SMArtCARE Registry Study
Abstract
Background and objectives: Disease progression in patients with spinal muscular atrophy (SMA) has changed dramatically within the past years due to the approval of three different disease-modifying treatments. Nusinersen was the first drug to be approved for the treatment of SMA patients. Clinical trials provided data from infants with SMA type 1 and children with SMA type 2, but there is still insufficient evidence and only scarcely reported long-term experience for nusinersen treatment in ambulant patients. Here, we report data from the SMArtCARE registry of ambulant patients under nusinersen treatment with a follow-up period of up to 38 months.
Methods: SMArtCARE is a disease-specific registry in Germany, Austria and Switzerland. Data are collected as real-world data during routine patient visits. Our analysis included all patients under treatment with nusinersen able to walk independently before start of treatment with focus on changes in motor function.
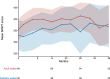
Results: Data from 231 ambulant patients were included in the analysis. During the observation period, 31 pediatric walkers (27.2%) and 31 adult walkers (26.5%) experienced a clinically meaningful improvement of≥30 m in the 6-Minute-Walk-Test. In contrast, only five adult walkers (7.7%) showed a decline in walking distance≥30 m, and two pediatric walkers (1.8%) lost the ability to walk unassisted under treatment with nusinersen. HFMSE and RULM scores improved in pediatric and remained stable in adult patients.
Conclusion: Our data demonstrate a positive effect of nusinersen treatment in most ambulant pediatric and adult SMA patients. We not only observed a stabilization of disease progression or lack of deterioration, but clinically meaningful improvements in walking distance.
Keywords: SMArtCARE; Spinal muscular atrophy; ambulant; nusinersen; walker.
Conflict of interest statement
A. Pechmann received compensation for advisory boards, training activities and research grants from Novartis and Biogen. M. Schimmel received compensation for advisory boards from Roche; A. Blaschek received personal fees for advisory board services / speakers honoraria from Novartis, Roche, Ad medicum, Sanofi Genzyme and Avexis; JC Koch received compensation for advisory boards and talks from Biogen and Roche; D. Zeller received compensation for scientific consultancy from Novartis, for advisory boards from Biogen, and for training activities from Desitin; M. Baumann received compensation for advisory boards and speakers honoraria from Novartis, Biogen and Roche; R. Trollmann reports personal fees from Desitin Pharma, Novartis, PTC Therapeutics; M. Weiler received compensation for advisory boards, consultant and speaker honoraria and/or financial support for conference attendances from Akcea Therapeutics, Alnylam Pharmaceuticals, Biogen, Pfizer, Roche, and Sobi; C. Neuwirth received compensation for advisory boards and training activities by Biogen, Roche, Genzyme and Mitsubishi Tanabe; J. Friese received compensation for an advisory board from Novartis Gene Therapies; B. Schrank received compensation for advisory board from Biogen; M. Flotats-Bastardas received compensation for advisory boards from Biogen, Novartis and Roche; M.C. Walter received compensation for advisory boards, funding for travel or speaker honoraria, and research support from Biogen, Novartis, and Roche; R. Günther received compensation for advisory boards and research support from Biogen and compensation for advisory boards from Hofmann La-Roche; M. Türk received compensation for a presentation and an advisory board from Sanofi Genzyme; T. Hagenacker received compensation for advisory board, lectures and research grants from Biogen, Roche and Novartis; J. Johansen received honoraria for advisory board participation and/or lectures and/or manuscript writing from Avexis/Novartis, Biogen, Roche, PTC, Pfizer and Sarepta Th., respectively, and financial support from Biogen for the SMArtCARE registry (paid to institution); S. Petri received compensation for advisory boards from Biogen, Roche and Zambon, speaker honoraria from Biogen, Roche, Italfarmaco and Novartis, grants from the German Israeli Foundation (GIF), German Neuromuscular Society (DGM) and Neurodegenerative Research (NDR); C. Stadler received fees for speeches & participation in discussions from Merz Pharma; B. Becker received financial support for a publication from Biogen; C. Kamm received honoraria as a speaker and advisory board member from Biogen, Roche and Ipsen; C. Köhler received honoraria from Roche, PTC, Biogen and Genzyme; M. Smitka received funding for educational activities and participation in advisory boards concering SMA from Novartis Gene Therapies, Biogen and Roche; H. Kölbel received compensation for presentations and consultancy from Avexis, Biogen, Sanofi, Pfizer, Novartis, and Roche; A. Ziegler receives honoraria for advisory boards and speaker honoraria from Novartis Gene Therapies, Roche Pharma and Biogen. His institution receives research grants from Biogen; G. Bernert receives research suppert from PTC and honoraria from Novartis Gene Therapies, PTC, Biogen, Roche, Pfizer, Santhera; E. Wilichowski received compensation for advisory boards from Biogen, Novartis and Roche; H. Lochmüller received consultancy and financial support for research projects and clinical trials from Amplo Biotechnology, AMO Pharma, argenx, Biogen, Desitin, Fulcrum Therapeutics, Harmony Biosciences, KYE Pharmaceuticals, Milo Biotechnology, Novartis, Pfizer, PTC Therapeutics, Hoffman-La Roche Limited, Sanofi-Genzyme, Santhera, Sarepta, Satellos, Spark Therapeutics and Ultragenyx. HL is the Editor-in-chief for the Journal of Neuromuscular Diseases (IOS Press); J. Kirschner received support from Biogen, Novartis, Roche and ScholarRock for consultancy, educational activities and/or clinical research.
Figures



References
-
- Lusakowska A, Jedrzejowska M, Kaminska A, Janiszewska K, Grochowski P, Zimowski J, et al.., Observation of the natural course of type 3 spinal muscular atrophy: Data from the polish registry of spinal muscular atrophy, Orphanet J Rare Dis. 2021;16:150. doi: 10.1186/s13023-021-01771-y. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

