Case report: Persistent shedding of a live vaccine-derived rubella virus in a young man with severe combined immunodeficiency and cutaneous granuloma
- PMID: 36569925
- PMCID: PMC9773200
- DOI: 10.3389/fimmu.2022.1075351
Case report: Persistent shedding of a live vaccine-derived rubella virus in a young man with severe combined immunodeficiency and cutaneous granuloma
Abstract
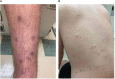
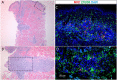
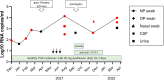
A young man with X-linked severe combined immunodeficiency developed a persistent vaccine-derived rubella virus (VDRV) infection, with the emergence of cutaneous granulomas more than fifteen years after receipt of two doses of measles-mumps-rubella (MMR) vaccine. Following nasopharyngeal swab (NP) collection, VDRV was detected by real-time polymerase chain reaction (RT-qPCR) and sequencing, and live, replication-competent VDRV was isolated in cell culture. To assess duration and intensity of viral shedding, sequential respiratory samples, one cerebrospinal fluid sample, and two urine samples were collected over 15 months, and VDRV RNA was detected in all samples by RT-qPCR. Live VDRV was cultured from nine of the eleven respiratory specimens and from one urine specimen. To our knowledge, this was the first reported instance of VDRV cultured from respiratory specimens or from urine. To assess potential transmission to close contacts, NP specimens and sera were collected from all household contacts, all of whom were immunocompetent and previously vaccinated with MMR. VDRV RNA was not detected in any NP swabs from the contacts, nor did serologic investigations suggest VDRV transmission to any contacts. This report highlights the need to understand the prevalence and duration of VDRV shedding in granuloma patients and to estimate the risk of VDRV transmission to immune and non-immune contacts.
Keywords: gene therapy; inborn errors of immunity; live virus shedding; skin granuloma; vaccine-derived rubella virus; viral persistence.
Copyright © 2022 Bonner, Sukerman, Liko, Lanzieri, Sutton, DeBess, Leesman, Icenogle, Hao, Chen, Faisthalab, Leman, Cieslak, DeRavin and Perelygina.
Conflict of interest statement
Author CL was employed by company Transformative Health and Wellness. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- Leung J, Sullivan KE, Perelygina L, Icenogle JP, Fuleihan RL, Lanzieri TM. Prevalence of granulomas in patients with primary immunodeficiency disorders, united states: Data from national health care claims and the US immunodeficiency network registry. J Clin Immunol (2018) 38(6):717–26. doi: 10.1007/s10875-018-0534-7 - DOI - PMC - PubMed
-
- Bodemer C, Sauvage V, Mahlaoui N, Cheval J, Couderc T, Leclerc-Mercier S, et al. . Live rubella virus vaccine long-term persistence as an antigenic trigger of cutaneous granulomas in patients with primary immunodeficiency. Clin Microbiol Infect (2014) 20(10):O656–63. doi: 10.1111/1469-0691.12573 - DOI - PubMed
-
- Perelygina L, Faisthalab R, Abernathy E, Chen M-H, Hao L, Bercovitch L, et al. . Rubella virus infected macrophages and neutrophils define patterns of granulomatous inflammation in inborn and acquired errors of immunity. Front Immunol (2021) 12:5452. doi: 10.3389/fimmu.2021.796065 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

