Spectral trends in GC-EI-MS data obtained from the SWGDRUG mass spectral library and literature: A resource for the identification of unknown compounds
- PMID: 36578315
- PMCID: PMC9793444
- DOI: 10.1016/j.forc.2022.100459
Spectral trends in GC-EI-MS data obtained from the SWGDRUG mass spectral library and literature: A resource for the identification of unknown compounds
Abstract
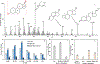
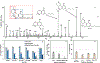
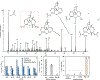
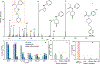
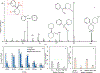
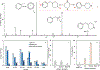
Rapid identification of new or emerging psychoactive substances remains a critical challenge in forensic drug chemistry laboratories. Current analytical protocols are well-designed for confirmation of known substances yet struggle when new compounds are encountered. Many laboratories initially attempt to classify new compounds using gas chromatography-electron ionization-mass spectrometry (GC-EI-MS). Though there is a large body of research focused on the analysis of illicit substances with GC-EI-MS, there is little high-level discussion of mass spectral trends for different classes of drugs. This manuscript compiles literature information and performs simple exploratory analyses on evaluated GC-EI-MS data to investigate mass spectral trends for illicit substance classes. Additionally, this work offers other important aspects: brief discussions of how each class of drugs is used; illustrations of EI mass spectra with proposed structures of commonly observed ions; and summaries of mass spectral trends that can help an analyst classify new illicit compounds.
Keywords: Drug trends; GC-EI-MS; Mass spectra commonalities; Novel psychoactive substances (NPS); Seized drugs.
Figures


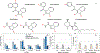




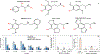


Similar articles
-
GC-EI-MS datasets of trimethylsilyl (TMS) and tert-butyl dimethyl silyl (TBDMS) derivatives for development of machine learning-based compound identification approaches.Data Brief. 2023 Apr 11;48:109138. doi: 10.1016/j.dib.2023.109138. eCollection 2023 Jun. Data Brief. 2023. PMID: 37128582 Free PMC article.
-
Comprehensive Data Evaluation Methods Used in Developing the SWGDRUG Mass Spectral Reference Library for Seized Drug Identification.Anal Chem. 2024 Oct 22;96(42):17004-17012. doi: 10.1021/acs.analchem.4c04425. Epub 2024 Oct 8. Anal Chem. 2024. PMID: 39378263
-
Sulfur organic compounds in bottom sediments of the eastern Gulf of Finland.Environ Sci Pollut Res Int. 2007 Sep;14(6):366-76. doi: 10.1065/espr2006.08.334. Environ Sci Pollut Res Int. 2007. PMID: 17993219
-
Fatal cases involving new psychoactive substances and trends in analytical techniques.Front Toxicol. 2022 Oct 25;4:1033733. doi: 10.3389/ftox.2022.1033733. eCollection 2022. Front Toxicol. 2022. PMID: 36387045 Free PMC article. Review.
-
Electron ionization mass spectrometry: Quo vadis?Electrophoresis. 2022 Aug;43(15):1587-1600. doi: 10.1002/elps.202100392. Epub 2022 May 20. Electrophoresis. 2022. PMID: 35531989 Review.
Cited by
-
Online and Offline Prioritization of Chemicals of Interest in Suspect Screening and Non-targeted Screening with High-Resolution Mass Spectrometry.Anal Chem. 2024 Mar 5;96(9):3707-3716. doi: 10.1021/acs.analchem.3c05705. Epub 2024 Feb 21. Anal Chem. 2024. PMID: 38380899 Free PMC article. Review.
-
Investigation of New Psychoactive Substances (NPS), Other Illicit Drugs, and Drug-Related Compounds in a Taiwanese Wastewater Sample Using High-Resolution Mass-Spectrometry-Based Targeted and Suspect Screening.Molecules. 2023 Jun 28;28(13):5040. doi: 10.3390/molecules28135040. Molecules. 2023. PMID: 37446702 Free PMC article.
-
PS2MS: A Deep Learning-Based Prediction System for Identifying New Psychoactive Substances Using Mass Spectrometry.Anal Chem. 2024 Mar 26;96(12):4835-4844. doi: 10.1021/acs.analchem.3c05019. Epub 2024 Mar 15. Anal Chem. 2024. PMID: 38488022 Free PMC article.
-
Classification of Mass Spectral Data to Assist in the Identification of Novel Synthetic Cannabinoids.Molecules. 2024 Sep 30;29(19):4646. doi: 10.3390/molecules29194646. Molecules. 2024. PMID: 39407576 Free PMC article.
-
On the challenge of unambiguous identification of fentanyl analogs: Exploring measurement diversity using standard reference mass spectral libraries.J Forensic Sci. 2023 Sep;68(5):1494-1503. doi: 10.1111/1556-4029.15322. Epub 2023 Jul 11. J Forensic Sci. 2023. PMID: 37431311 Free PMC article.
References
-
- Lukić V, Micić R, Arsić B, Nedović B, Radosavljević Ž, Overview of the major classes of new psychoactive substances, psychoactive effects, analytical determination and conformational analysis of selected illegal drugs, Open Chem. 19 (2021) 60–106, 10.1515/chem-2021-0196. - DOI
-
- Strano Rossi S, Odoardi S, Gregori A, Peluso G, Ripani L, Ortar G, Serpelloni G, Romolo FS, An analytical approach to the forensic identification of different classes of new psychoactive substances (NPSs) in seized materials, Rapid Commun. Mass Spectrom 28 (2014) 1904–1916, 10.1002/rcm.6969. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
