A Novel Murine Model of Endothelial Keratoplasty
- PMID: 36582035
- PMCID: PMC9805546
- DOI: 10.1097/ICO.0000000000003047
A Novel Murine Model of Endothelial Keratoplasty
Abstract
Purpose: The purpose of this study was to establish a murine model of endothelial keratoplasty.
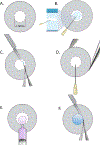
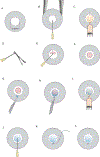
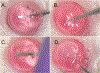
Methods: Endothelial keratoplasty (EK) was performed using C57BL/6 donor and BALB/c recipient mice. The central endothelium and Descemet membrane were removed from the recipient cornea, and a 1.5-mm posterior lamellar donor graft was made adherent to the recipient cornea with a small amount of viscoelastic. Mice were followed through slitlamp microscopy postoperatively, and OCT was used to assess the cornea and anterior chamber and measure central corneal thickness. Histology and immunohistochemistry were performed to confirm graft adherence and endothelial cell morphology.
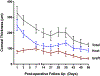
Results: Successfully attached EK grafts were visualized in all transplanted animals. Histology and immunostaining confirmed proper graft orientation and adherence, as well as the presence of donor endothelium on transplanted grafts. We observed maximal corneal edema in all animals at day 1 postoperatively which gradually subsided. EK graft survival was 97% at 8 weeks.
Conclusions: In this study, we describe a novel murine model for EK which we anticipate will enable detailed investigation into the cellular and molecular mechanisms involved in EK pathobiology.
Copyright © 2022 Wolters Kluwer Health, Inc. All rights reserved.
Conflict of interest statement
The authors have no conflicts of interest to disclose.
Figures



References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

