Cyclooxygenase-2 activates EGFR-ERK1/2 pathway via PGE2-mediated ADAM-17 signaling in testosterone-induced benign prostatic hyperplasia
- PMID: 36586043
- PMCID: PMC9958186
- DOI: 10.1007/s10787-022-01123-7
Cyclooxygenase-2 activates EGFR-ERK1/2 pathway via PGE2-mediated ADAM-17 signaling in testosterone-induced benign prostatic hyperplasia
Abstract
Objective and design: Prostatic inflammation is the driving force in benign prostatic hyperplasia (BPH). This work investigated the potential modulatory effect of COX-2 inhibition on ADAM-17/EGFR/ERK1/2 axis.
Materials or subjects: Adult male Wistar rats were used.
Treatment: Celecoxib (10 and 20 mg/kg; i.p.) was injected i.p. daily for three weeks. Testosterone (TST) (3 mg/kg; s.c.) was used to induce BPH.
Methods: Prostatic inflammation and hyperplasia were assessed by organ weight and histopathology. Inflammatory mediators were measured using ELISA technique. Protein analysis was performed using western blotting and immunohistochemistry. Gene expression analysis was performed using qRT-PCR. Statistical analyses included one-way ANOVA and Tukey's multiple comparison test.
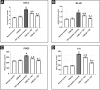
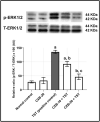
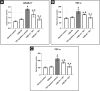
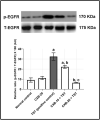
Results: Testosterone-treated rats had a marked increase in COX-2, prostate weight, and index. Moreover, TST-induced COX-2 was inferred from cytoskeletal changes and was attributable to the overexpression of PGE2, NF-κB (p65), and IL-6. COX-2-derived PGE2 increased the activity of ADAM-17, TGF-α, and TNF-α. Consequently, EGFR-ERK1/2 pathway was over-activated, disrupting anti-apoptotic Bcl-2, cyclin D1, and pro-apoptotic Bax. Celecoxib reversed these effects.
Conclusion: COX-2 stimulates the ERK1/2 pathway via PGE2-ADAM-17-catalyzed shedding of TGF-α in testosterone-induced BPH. The results indicate a functional correlation between inflammation and hyperplasia in BPH.
Keywords: ADAM-17 (TACE); Benign prostatic hyperplasia; COX-2; Celecoxib; EGFR–ERK1/2; PGE2.
© 2022. The Author(s).
Conflict of interest statement
The authors have no relevant financial or non-financial interests to disclose.
Figures



Similar articles
-
Probenecid ameliorates testosterone-induced benign prostatic hyperplasia: Implications of PGE-2 on ADAM-17/EGFR/ERK1/2 signaling cascade.J Biochem Mol Toxicol. 2023 Nov;37(11):e23450. doi: 10.1002/jbt.23450. Epub 2023 Jun 23. J Biochem Mol Toxicol. 2023. PMID: 37352135
-
Common salt aggravated pathology of testosterone-induced benign prostatic hyperplasia in adult male Wistar rat.BMC Urol. 2023 Dec 11;23(1):207. doi: 10.1186/s12894-023-01371-x. BMC Urol. 2023. PMID: 38082261 Free PMC article.
-
Iodine prevents the increase of testosterone-induced oxidative stress in a model of rat prostatic hyperplasia.Free Radic Biol Med. 2018 Feb 1;115:298-308. doi: 10.1016/j.freeradbiomed.2017.12.014. Epub 2017 Dec 15. Free Radic Biol Med. 2018. PMID: 29248723
-
Curcuma oil ameliorates benign prostatic hyperplasia through suppression of the nuclear factor-kappa B signaling pathway in rats.J Ethnopharmacol. 2021 Oct 28;279:113703. doi: 10.1016/j.jep.2020.113703. Epub 2020 Dec 17. J Ethnopharmacol. 2021. PMID: 33340599 Free PMC article.
-
Traditional Chinese Medicine Danzhi qing'e decoction inhibits inflammation-associated prostatic hyperplasia via inactivation of ERK1/2 signal pathway.J Ethnopharmacol. 2023 Jun 12;309:116354. doi: 10.1016/j.jep.2023.116354. Epub 2023 Mar 9. J Ethnopharmacol. 2023. PMID: 36906158
Cited by
-
The pathogenesis of benign prostatic hyperplasia and the roles of Prdx3, oxidative stress, pyroptosis and autophagy:a review.Front Oncol. 2025 Aug 5;15:1579539. doi: 10.3389/fonc.2025.1579539. eCollection 2025. Front Oncol. 2025. PMID: 40837016 Free PMC article. Review.
-
Ubiquinol attenuates γ-radiation induced coronary and aortic changes via PDGF/p38 MAPK/ICAM-1 related pathway.Sci Rep. 2023 Dec 27;13(1):22959. doi: 10.1038/s41598-023-50218-w. Sci Rep. 2023. PMID: 38151557 Free PMC article.
-
Unveiling therapeutic targets and preventive components for kidney insufficiency and blood stasis-type BPH: bridging metabolomics, network pharmacology and reverse screening.Front Pharmacol. 2025 Jun 19;16:1584766. doi: 10.3389/fphar.2025.1584766. eCollection 2025. Front Pharmacol. 2025. PMID: 40626307 Free PMC article.
-
Nanomedicine in HNSCC therapy-a challenge to conventional therapy.Front Pharmacol. 2024 Oct 14;15:1434994. doi: 10.3389/fphar.2024.1434994. eCollection 2024. Front Pharmacol. 2024. PMID: 39469621 Free PMC article. Review.
-
Diacerein provokes apoptosis, improves redox balance, and downregulates PCNA and TNF-α in a rat model of testosterone-induced benign prostatic hyperplasia: A new non-invasive approach.PLoS One. 2023 Nov 9;18(11):e0293682. doi: 10.1371/journal.pone.0293682. eCollection 2023. PLoS One. 2023. PMID: 37943844 Free PMC article.
References
-
- Abo-El Fetoh ME, Helal GK, Saleh IG, Ewees M, Elshafey M, Elnagar MR, Akool E-S. Cyclosporin A activates human hepatocellular carcinoma (HepG2 cells) proliferation: implication of EGFR-mediated ERK1/2 signaling pathway. Naunyn Schmiedebergs Arch Pharmacol. 2020;393:897–908. doi: 10.1007/s00210-019-01798-w. - DOI - PubMed
-
- Afify H, Abo-Youssef AM, Abdel-Rahman HM, Allam S, Azouz AA. The modulatory effects of cinnamaldehyde on uric acid level and IL-6/JAK1/STAT3 signaling as a promising therapeutic strategy against benign prostatic hyperplasia. Toxicol Appl Pharmacol. 2020;402:115122. doi: 10.1016/j.taap.2020.115122. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous
