The adenosine A2A receptor antagonist KW6002 distinctly regulates retinal ganglion cell morphology during postnatal development and neonatal inflammation
- PMID: 36588710
- PMCID: PMC9800499
- DOI: 10.3389/fphar.2022.1082997
The adenosine A2A receptor antagonist KW6002 distinctly regulates retinal ganglion cell morphology during postnatal development and neonatal inflammation
Abstract
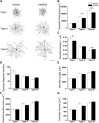
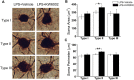
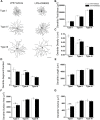
Adenosine A2A receptors (A2ARs) appear early in the retina during postnatal development, but the roles of the A2ARs in the morphogenesis of distinct types of retinal ganglion cells (RGCs) during postnatal development and neonatal inflammatory response remain undetermined. As the RGCs are rather heterogeneous in morphology and functions in the retina, here we resorted to the Thy1-YFPH transgenic mice and three-dimensional (3D) neuron reconstruction to investigate how A2ARs regulate the morphogenesis of three morphologically distinct types of RGCs (namely Type I, II, III) during postnatal development and neonatal inflammation. We found that the A2AR antagonist KW6002 did not change the proportion of the three RGC types during retinal development, but exerted a bidirectional effect on dendritic complexity of Type I and III RGCs and cell type-specifically altered their morphologies with decreased dendrite density of Type I, decreased the dendritic field area of Type II and III, increased dendrite density of Type III RGCs. Moreover, under neonatal inflammation condition, KW6002 specifically increased the proportion of Type I RGCs with enhanced the dendrite surface area and volume and the proportion of Type II RGCs with enlarged the soma area and perimeter. Thus, A2ARs exert distinct control of RGC morphologies to cell type-specifically fine-tune the RGC dendrites during normal development but to mainly suppress RGC soma and dendrite volume under neonatal inflammation.
Keywords: 3D reconstruction; adenosine A2A receptor; development; morphology; neonatal inflammation; retinal ganglion cell.
Copyright © 2022 Hu, Li, Zhang, Shi, Tang, Zhang, Kuang, Chen, Qu and Gao.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures





Similar articles
-
Neonatal inflammation induces reorganization in dendritic morphology of retinal ganglion cells but not their retinogeniculate projection in mice.Neurosci Lett. 2018 May 29;676:34-40. doi: 10.1016/j.neulet.2018.04.012. Epub 2018 Apr 5. Neurosci Lett. 2018. PMID: 29627341
-
Effects of early postnatal exposure to ethanol on retinal ganglion cell morphology and numbers of neurons in the dorsolateral geniculate in mice.Alcohol Clin Exp Res. 2011 Nov;35(11):2063-74. doi: 10.1111/j.1530-0277.2011.01557.x. Epub 2011 Jun 8. Alcohol Clin Exp Res. 2011. PMID: 21651582 Free PMC article.
-
Selective Vulnerability of Specific Retinal Ganglion Cell Types and Synapses after Transient Ocular Hypertension.J Neurosci. 2016 Aug 31;36(35):9240-52. doi: 10.1523/JNEUROSCI.0940-16.2016. J Neurosci. 2016. PMID: 27581463 Free PMC article.
-
Retinal ganglion cells: Energetics, compartmentation, axonal transport, cytoskeletons and vulnerability.Prog Retin Eye Res. 2013 Sep;36:217-46. doi: 10.1016/j.preteyeres.2013.07.001. Epub 2013 Jul 23. Prog Retin Eye Res. 2013. PMID: 23891817 Review.
-
Retinal ganglion cell dendritic development and its control. Filling the gaps.Mol Neurobiol. 1996 Apr;12(2):133-44. doi: 10.1007/BF02740650. Mol Neurobiol. 1996. PMID: 8818147 Review.
Cited by
-
Pharmacology of Adenosine Receptors: Recent Advancements.Biomolecules. 2023 Sep 14;13(9):1387. doi: 10.3390/biom13091387. Biomolecules. 2023. PMID: 37759787 Free PMC article. Review.
References
-
- Aires I. D., Boia R., Rodrigues-Neves A. C., Madeira M. H., Marques C., Ambrosio A. F., et al. (2019a). Blockade of microglial adenosine A2A receptor suppresses elevated pressure-induced inflammation, oxidative stress, and cell death in retinal cells. Glia 67 (5), 896–914. 10.1002/glia.23579 - DOI - PMC - PubMed
-
- Aires I. D., Madeira M. H., Boia R., Rodrigues-Neves A. C., Martins J. M., Ambrosio A. F., et al. (2019b). Intravitreal injection of adenosine A(2A) receptor antagonist reduces neuroinflammation, vascular leakage and cell death in the retina of diabetic mice. Sci. Rep. 9, 17207. 10.1038/s41598-019-53627-y - DOI - PMC - PubMed
-
- Alcada-Morais S., Goncalves N., Moreno-Juan V., Andres B., Ferreira S., Marques J. M., et al. (2021). Adenosine A2A receptors contribute to the radial migration of cortical projection neurons through the regulation of neuronal polarization and axon formation. Cereb. Cortex 31 (12), 5652–5663. 10.1093/cercor/bhab188 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

