Meta‑analysis of the efficacy and safety of nifekalant in the conversion of atrial fibrillation
- PMID: 36588807
- PMCID: PMC9780521
- DOI: 10.3892/etm.2022.11755
Meta‑analysis of the efficacy and safety of nifekalant in the conversion of atrial fibrillation
Abstract
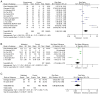
Atrial fibrillation (AF) is the most common type of supraventricular tachyarrhythmia. Nifekalant is a new class III antiarrhythmic drug approved for the treatment of ventricular tachyarrhythmias, but its effectiveness in converting AF to sinus rhythm remains unclear. The present analysis aimed to investigate the effect of nifekalant in the conversion of AF. PubMed, Cochrane Library and China National Knowledge Infrastructure databases were systematically used to search relevant studies published between 1999 (data at which the drug was first approved for marketing in Japan) and 2022. Randomized clinical trials, prospective studies and retrospective studies on the use of nifekalant for AF were screened. The study metrics included the success rate of the conversion of AF, the mean time to conversion, the success rate of 12 months after a single AF catheter ablation procedure and the incidence of adverse events. The eligible studies screened included six randomized clinical trials, three prospective studies and three retrospective studies, totalling 12 studies with 1,162 patients. The risk ratio (RR) for successful conversion in the nifekalant and control groups was 1.95 [95% confidence interval (CI), 1.23-3.08; P=0.005] and the mean difference for the mean time to conversion was -1.73 [95% CI, -2.69-(-0.77); P=0.0004]. Statistically significant differences were observed between nifekalant and control groups. Subgroup analysis revealed a statistically significant difference in the success rate of conversion following catheter ablation in the nifekalant group compared with the amiodarone group and the RR value was 1.95 (95% CI, 1.37-2.77; P=0.0002). Statistically significant difference was observed compared with the electrical cardioversion group and the RR value was 0.90 (95% CI, 0.84-0.98; P=0.01). However, the combined RR values for the two groups were 1.18 (95% CI, 0.85-1.65; P<0.0002). The RR value for adverse events was 0.85 (95% CI, 0.51-1.43; P=0.55), with no statistically significant differences between nifekalant and control groups. In conclusion, the results demonstrated that the success rate and time to conversion in the nifekalant group were improved compared with those in the control group, particularly after catheter ablation, and the conversion effect with nifekalant was significantly improved compared with that in the control group.
Keywords: amiodarone; atrial fibrillation; nifekalant; radiofrequency ablation.
Copyright: © Liu et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
-
- Yandrapalli S, Malik AH, Namrata F, Pemmasani G, Bandyopadhyay D, Vallabhajosyula S, Aronow WS, Frishman WH, Jain D, Cooper HA, Panza JA. Influence of diabetes mellitus interactions with cardiovascular risk factors on post-myocardial infarction heart failure hospitalizations. Int J Cardiol. 2022;348:140–146. doi: 10.1016/j.ijcard.2021.11.086. - DOI - PubMed
LinkOut - more resources
Full Text Sources
