PEO Coatings Modified with Halloysite Nanotubes: Composition, Properties, and Release Performance
- PMID: 36613748
- PMCID: PMC9820610
- DOI: 10.3390/ijms24010305
PEO Coatings Modified with Halloysite Nanotubes: Composition, Properties, and Release Performance
Abstract
In this work, the properties of the coatings formed on the Mg-Mn-Ce alloy by plasma electrolytic oxidation (PEO) in electrolytes containing halloysite nanotubes (HNTs) were investigated. The incorporation of halloysite nanotubes into the PEO coatings improved their mechanical characteristics, increased thickness, and corrosion resistance. The studied layers reduced corrosion current density by more than two times in comparison with the base PEO layer without HNTs (from 1.1 × 10-7 A/cm2 to 4.9 × 10-8 A/cm2). The presence of halloysite nanotubes and products of their dihydroxylation that were formed under the PEO conditions had a positive impact on the microhardness of the obtained layers (this parameter increased from 4.5 ± 0.4 GPa to 7.3 ± 0.5 GPa). In comparison with the base PEO layer, coatings containing halloysite nanotubes exhibited sustained release and higher adsorption capacity regarding caffeine.
Keywords: caffeine release; halloysite nanotubes; multifunctional coatings; plasma electrolytic oxidation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




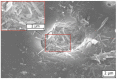





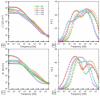





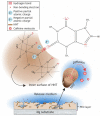
References
-
- Kulekci M.K. Magnesium and Its Alloys Applications in Automotive Industry. Int. J. Adv. Manuf. Technol. 2008;39:851–865. doi: 10.1007/s00170-007-1279-2. - DOI
-
- Song J., She J., Chen D., Pan F. Latest Research Advances on Magnesium and Magnesium Alloys Worldwide. J. Magnes. Alloy. 2020;8:1–41. doi: 10.1016/j.jma.2020.02.003. - DOI
-
- Furuya H., Kogiso N., Matunaga S., Senda K. Applications of Magnesium Alloys for Aerospace Structure Systems. Mater. Sci. Forum. 2000;350–351:341–348.
-
- Joost W.J., Krajewski P.E. Towards Magnesium Alloys for High-Volume Automotive Applications. Scr. Mater. 2017;128:107–112. doi: 10.1016/j.scriptamat.2016.07.035. - DOI
-
- Śliwa R.E., Balawender T., Hadasik E., Kuc D., Gontarz A., Korbel A., Bochniak W. Metal Forming of Lightweight Magnesium Alloys for Aviation Applications. Arch. Metall. Mater. 2017;62:1559–1566. doi: 10.1515/amm-2017-0239. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

