Determination of Hg(II) and Methylmercury by Electrothermal Atomic Absorption Spectrometry after Dispersive Solid-Phase Microextraction with a Graphene Oxide Magnetic Material
- PMID: 36615211
- PMCID: PMC9822199
- DOI: 10.3390/molecules28010014
Determination of Hg(II) and Methylmercury by Electrothermal Atomic Absorption Spectrometry after Dispersive Solid-Phase Microextraction with a Graphene Oxide Magnetic Material
Abstract
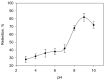
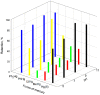
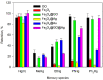
The toxicity of all species of mercury makes it necessary to implement analytical procedures capable of quantifying the different forms this element presents in the environment, even at very low concentrations. In addition, due to the assorted environmental and health consequences caused by each mercury species, it is desirable that the procedures are able to distinguish these forms. In nature, mercury is mainly found as Hg0, Hg2+ and methylmercury (MeHg), with the latter being rapidly assimilated by living organisms in the aquatic environment and biomagnified through the food chain. In this work, a dispersive solid-phase microextraction of Hg2+ and MeHg is proposed using as the adsorbent a magnetic hybrid material formed by graphene oxide and ferrite (Fe3O4@GO), along with a subsequent determination by electrothermal atomic absorption spectrometry (ETAAS). On the one hand, when dithizone at a pH = 5 is used as an auxiliary agent, both Hg(II) and MeHg are retained on the adsorbent. Next, for the determination of both species, the solid collected by the means of a magnet is suspended in a mixture of 50 µL of HNO3 (8% v/v) and 50 µL of H2O2 at 30% v/v by heating for 10 min in an ultrasound thermostatic bath at 80 °C. On the other hand, when the sample is set at a pH = 9, Hg(II) and MeHg are also retained, but if the solid collected is washed with N-acetyl-L-cysteine only, then the Hg(II) remains on the adsorbent, and can be determined as indicated above. The proposed procedure exhibits an enrichment factor of 49 and the determination presents a linear range between 0.1 and 10 µg L-1 of mercury. The procedure has been applied to the determination of mercury in water samples from different sources.
Keywords: electrothermal atomic absorption spectrometry; magnetic dispersive solid-phase microextraction; mercury; methylmercury; speciation.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures

Similar articles
-
Speciation of mercury in water and biological samples by eco-friendly ultrasound-assisted deep eutectic solvent based on liquid phase microextraction with electrothermal atomic absorption spectrometry.Talanta. 2019 May 15;197:310-318. doi: 10.1016/j.talanta.2019.01.018. Epub 2019 Jan 9. Talanta. 2019. PMID: 30771941
-
Selective measurement of ultratrace methylmercury in fish by flow injection on-line microcolumn displacement sorption preconcentration and separation coupled with electrothermal atomic absorption spectrometry.Anal Chem. 2003 May 15;75(10):2251-5. doi: 10.1021/ac026415f. Anal Chem. 2003. PMID: 12918963
-
Using a Simple Magnetic Adsorbent for the Preconcentration and Determination of Ga(III) and In(III) by Electrothermal Atomic Absorption Spectrometry.Molecules. 2023 Mar 10;28(6):2549. doi: 10.3390/molecules28062549. Molecules. 2023. PMID: 36985521 Free PMC article.
-
Magnetic solid-phase extraction based on sulfur-functionalized magnetic metal-organic frameworks for the determination of methylmercury and inorganic mercury in water and fish samples.J Chromatogr A. 2021 Sep 27;1654:462465. doi: 10.1016/j.chroma.2021.462465. Epub 2021 Aug 12. J Chromatogr A. 2021. PMID: 34416446
-
Solid phase extraction for the speciation and preconcentration of inorganic selenium in water samples: a review.Anal Chim Acta. 2013 Dec 4;804:37-49. doi: 10.1016/j.aca.2013.09.054. Epub 2013 Oct 5. Anal Chim Acta. 2013. PMID: 24267061 Review.
Cited by
-
Effect of Organic Solvents on the Activity, Stability and Secondary Structure of asclepain cI, Using FTIR and Molecular Dynamics Simulations.Protein J. 2024 Jun;43(3):487-502. doi: 10.1007/s10930-024-10182-4. Epub 2024 Mar 7. Protein J. 2024. PMID: 38453735
References
-
- Report, Utilizing Direct Mercury Analysis for Mercury Detection in Botanical Extracts, Vitamins, Minerals and Dietary Supplements. Braz. J. Anal. Chem. 2021;8:237–240.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

