Daratumumab for the treatment of refractory ANCA-associated vasculitis
- PMID: 36627149
- PMCID: PMC9835944
- DOI: 10.1136/rmdopen-2022-002742
Daratumumab for the treatment of refractory ANCA-associated vasculitis
Abstract
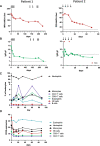
Objective Treatment-refractory antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV) is a life-threatening condition without evidence-based treatment options. One emerging treatment option for several antibody-mediated autoimmune diseases is the anti-CD38 antibody daratumumab, which depletes autoantibody-secreting plasma cells.Methods We treated two patients with severe life-threatening AAV with renal and pulmonary manifestation despite induction therapy with rituximab and cyclophosphamide with four to eight doses of 1800 mg daratumumab. We followed clinical and immunological responses.Results The first patient with myeloperoxidase-ANCA-positive microscopic polyangiitis had resolution of pneumonitis and pleuritis and stabilisation of kidney function after daratumumab. The second patient with proteinase 3-ANCA-positive granulomatosis with polyangiitis, diffuse alveolar haemorrhage necessitating extracorporeal membrane oxygenation (ECMO) and acute kidney failure, requiring kidney replacement therapy, was weaned off ECMO, mechanical ventilation and dialysis and discharged home after daratumumab. Clinical improvement was paralleled by a strong reduction in serum ANCA levels as well as total IgG, indicating depletion of plasma cells. Apart from the depletion of CD38+ natural killer cells, blood leucocyte levels were not notably influenced by daratumumab. Only mild adverse events, such as hypogammaglobulinaemia and an upper respiratory tract infection occurred.Conclusion Daratumumab was safe and effective in inducing remission in two patients with severe treatment-refractory AAV, warranting prospective clinical trials to establish safety and efficacy.
Keywords: Autoimmune Diseases; Systemic vasculitis; Treatment.
© Author(s) (or their employer(s)) 2023. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: None declared.
Figures


References
-
- Alexander T, Thiel A, Rosen O, et al. . Depletion of autoreactive immunologic memory followed by autologous hematopoietic stem cell transplantation in patients with refractory SLE induces long-term remission through de novo generation of a juvenile and tolerant immune system. Blood 2009;113:214–23. 10.1182/blood-2008-07-168286 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
