Contraception in breast cancer survivors from the FEERIC case-control study (performed on behalf of the Seintinelles research network)
- PMID: 36630821
- PMCID: PMC9982267
- DOI: 10.1016/j.breast.2022.12.033
Contraception in breast cancer survivors from the FEERIC case-control study (performed on behalf of the Seintinelles research network)
Abstract
Objective: To compare the prevalence of contraception in breast cancer (BC) patients at risk of unintentional pregnancy (i.e. not currently pregnant or trying to get pregnant) and matched controls.
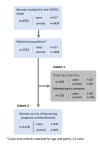
Study design: The FEERIC study (Fertility, Pregnancy, Contraception after BC in France) is a prospective, multicenter case-control study, including localized BC patients aged 18-43 years, matched for age and parity to cancer-free volunteer controls in a 1:2 ratio. Data were collected through online questionnaires completed on the Seintinelles research platform.
Results: In a population of 1278 women at risk of unintentional pregnancy, the prevalence of contraception at study inclusion did not differ significantly between cases (340/431, 78.9%) and controls (666/847, 78.6%, p = 0.97). Contrarily, the contraceptive methods used were significantly different, with a higher proportion of copper IUD use in BC survivors (59.5% versus 25.0% in controls p < 0.001). For patients at risk of unintentional pregnancy, receiving information about chemotherapy-induced ovary damage at BC diagnosis (OR = 2.47 95%CI [ 1.39-4.37] and anti-HER2 treatment (OR = 2.46, 95% CI [ 1.14-6.16]) were significantly associated with the use of a contraception in multivariate analysis.
Conclusion: In this large French study, BC survivors had a prevalence of contraception use similar to that for matched controls, though almost one in five women at risk of unintentional pregnancy did not use contraception. Dedicated consultations at cancer care centers could further improve access to information and contraception counseling.
Keywords: Breast cancer; Contraceptive counseling; Emergency contraception; Survivorship; contraception.
Copyright © 2022. Published by Elsevier Ltd.
Figures


References
-
- Defossez G., Le Guyader-Peyrou S., Uhry Z., Grosclaude P., Remontet L., Colonna M., et al. Saint‐Maurice: Santé publique France; 2019. Estimations nationales de l’incidence et de la mortalité par cancer en France métropolitaine entre 1990 et 2018. Étude à partir des registres des cancers du réseau Francim.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

