Accumulation of retrotransposons contributes to W chromosome differentiation in the willow beauty Peribatodes rhomboidaria (Lepidoptera: Geometridae)
- PMID: 36631492
- PMCID: PMC9834309
- DOI: 10.1038/s41598-023-27757-3
Accumulation of retrotransposons contributes to W chromosome differentiation in the willow beauty Peribatodes rhomboidaria (Lepidoptera: Geometridae)
Abstract
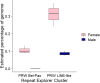
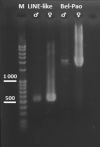
The W chromosome of Lepidoptera is typically gene-poor, repeat-rich and composed of heterochromatin. Pioneering studies investigating this chromosome reported an abundance of mobile elements. However, the actual composition of the W chromosome varies greatly between species, as repeatedly demonstrated by comparative genomic hybridization (CGH) or fluorescence in situ hybridization (FISH). Here we present an analysis of repeats on the W chromosome in the willow beauty, Peribatodes rhomboidaria (Geometridae), a species in which CGH predicted an abundance of W-enriched or W-specific sequences. Indeed, comparative analysis of male and female genomes using RepeatExplorer identified ten putative W chromosome-enriched repeats, most of which are LTR or LINE mobile elements. We analysed the two most abundant: PRW LINE-like and PRW Bel-Pao. The results of FISH mapping and bioinformatic analysis confirmed their enrichment on the W chromosome, supporting the hypothesis that mobile elements are the driving force of W chromosome differentiation in Lepidoptera. As the W chromosome is highly underrepresented in chromosome-level genome assemblies of Lepidoptera, this recently introduced approach, combining bioinformatic comparative genome analysis with molecular cytogenetics, provides an elegant tool for studying this elusive and rapidly evolving part of the genome.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

