Mechano-responsive hydrogel for direct stem cell manufacturing to therapy
- PMID: 36632503
- PMCID: PMC9817177
- DOI: 10.1016/j.bioactmat.2022.12.019
Mechano-responsive hydrogel for direct stem cell manufacturing to therapy
Abstract
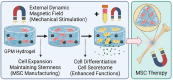
Bone marrow-derived mesenchymal stem cell (MSC) is one of the most actively studied cell types due to its regenerative potential and immunomodulatory properties. Conventional cell expansion methods using 2D tissue culture plates and 2.5D microcarriers in bioreactors can generate large cell numbers, but they compromise stem cell potency and lack mechanical preconditioning to prepare MSC for physiological loading expected in vivo. To overcome these challenges, in this work, we describe a 3D dynamic hydrogel using magneto-stimulation for direct MSC manufacturing to therapy. With our technology, we found that dynamic mechanical stimulation (DMS) enhanced matrix-integrin β1 interactions which induced MSCs spreading and proliferation. In addition, DMS could modulate MSC biofunctions including directing MSC differentiation into specific lineages and boosting paracrine activities (e.g., growth factor secretion) through YAP nuclear localization and FAK-ERK pathway. With our magnetic hydrogel, complex procedures from MSC manufacturing to final clinical use, can be integrated into one single platform, and we believe this 'all-in-one' technology could offer a paradigm shift to existing standards in MSC therapy.
Keywords: Cell therapy; DMS, Dynamic Mechanical Stimulation; Dynamic mechanical stimulation; MP, Magnetic Particle; MSC, Mesenchymal Stem Cell; Magnetic hydrogel; Mesenchymal stem cell; Stem cell manufacturing.
© 2023 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures







Similar articles
-
Comparability exercise of critical quality attributes of clinical-grade human mesenchymal stromal cells from the Wharton's jelly: single-use stirred tank bioreactors versus planar culture systems.Cytotherapy. 2024 May;26(5):418-426. doi: 10.1016/j.jcyt.2023.08.008. Epub 2023 Sep 16. Cytotherapy. 2024. PMID: 37715777
-
Hydrogel Culture Surface Stiffness Modulates Mesenchymal Stromal Cell Secretome and Alters Senescence.Tissue Eng Part A. 2020 Dec;26(23-24):1259-1271. doi: 10.1089/ten.tea.2020.0030. Epub 2020 Jul 6. Tissue Eng Part A. 2020. PMID: 32628570
-
Effect of 2D and 3D Culture Microenvironments on Mesenchymal Stem Cell-Derived Extracellular Vesicles Potencies.Front Cell Dev Biol. 2022 Feb 14;10:819726. doi: 10.3389/fcell.2022.819726. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35237601 Free PMC article.
-
Increased Mesenchymal Stem Cell Functionalization in Three-Dimensional Manufacturing Settings for Enhanced Therapeutic Applications.Front Bioeng Biotechnol. 2021 Feb 12;9:621748. doi: 10.3389/fbioe.2021.621748. eCollection 2021. Front Bioeng Biotechnol. 2021. PMID: 33644016 Free PMC article. Review.
-
Native and solubilized decellularized extracellular matrix: A critical assessment of their potential for improving the expansion of mesenchymal stem cells.Acta Biomater. 2017 Jun;55:1-12. doi: 10.1016/j.actbio.2017.04.014. Epub 2017 Apr 12. Acta Biomater. 2017. PMID: 28412553 Review.
Cited by
-
Multiscale Control of Nanofiber-Composite Hydrogel for Complex 3D Cell Culture by Extracellular Matrix Composition and Nanofiber Alignment.Biomater Res. 2024 May 29;28:0032. doi: 10.34133/bmr.0032. eCollection 2024. Biomater Res. 2024. PMID: 38812742 Free PMC article.
-
Dynamic Stimulations with Bioengineered Extracellular Matrix-Mimicking Hydrogels for Mechano Cell Reprogramming and Therapy.Adv Sci (Weinh). 2023 Jul;10(21):e2300670. doi: 10.1002/advs.202300670. Epub 2023 Apr 29. Adv Sci (Weinh). 2023. PMID: 37119518 Free PMC article. Review.
-
Acceleration of burn wound healing by micronized amniotic membrane seeded with umbilical cord-derived mesenchymal stem cells.Mater Today Bio. 2023 Jun 7;20:100686. doi: 10.1016/j.mtbio.2023.100686. eCollection 2023 Jun. Mater Today Bio. 2023. PMID: 37334186 Free PMC article.
-
Photonic control of ligand nanospacing in self-assembly regulates stem cell fate.Bioact Mater. 2023 Dec 26;34:164-180. doi: 10.1016/j.bioactmat.2023.12.011. eCollection 2024 Apr. Bioact Mater. 2023. PMID: 38343773 Free PMC article.
-
A bio-instructive, bioactive in situ polymerizable wound matrix promotes scar-free burn wound repair.iScience. 2025 Apr 17;28(5):112471. doi: 10.1016/j.isci.2025.112471. eCollection 2025 May 16. iScience. 2025. PMID: 40454101 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

