Dance with spins: site-directed spin labeling coupled to electron paramagnetic resonance spectroscopy directly inside cells
- PMID: 36633152
- PMCID: PMC9890500
- DOI: 10.1039/d2cc05907j
Dance with spins: site-directed spin labeling coupled to electron paramagnetic resonance spectroscopy directly inside cells
Abstract
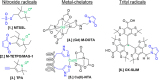
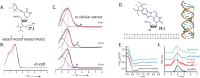
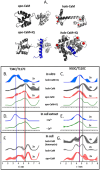
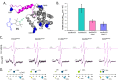
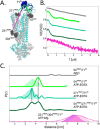
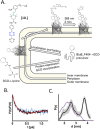
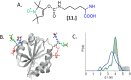
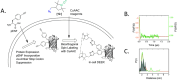
Depicting how biomolecules move and interact within their physiological environment is one of the hottest topics of structural biology. This Feature Article gives an overview of the most recent advances in Site-directed Spin Labeling coupled to Electron Paramagnetic Resonance spectroscopy (SDSL-EPR) to study biomolecules in living cells. The high sensitivity, the virtual absence of background, and the versatility of spin-labeling strategies make this approach one of the most promising techniques for the study of biomolecules in physiologically relevant environments. After presenting the milestones achieved in this field, we present a summary of the future goals and ambitions of this community.
Conflict of interest statement
There are no conflicts to declare.
Figures



Similar articles
-
In-Cell EPR: Progress towards Structural Studies Inside Cells.Chembiochem. 2020 Feb 17;21(4):451-460. doi: 10.1002/cbic.201900291. Epub 2019 Sep 26. Chembiochem. 2020. PMID: 31245902 Review.
-
Site-directed spin-labeling strategies and electron paramagnetic resonance spectroscopy for large riboswitches.Methods Enzymol. 2014;549:287-311. doi: 10.1016/B978-0-12-801122-5.00013-1. Methods Enzymol. 2014. PMID: 25432754
-
Elucidating the design principles of photosynthetic electron-transfer proteins by site-directed spin labeling EPR spectroscopy.Biochim Biophys Acta. 2016 May;1857(5):548-556. doi: 10.1016/j.bbabio.2015.08.009. Epub 2015 Sep 1. Biochim Biophys Acta. 2016. PMID: 26334844 Review.
-
Site directed spin labeling to elucidating the mechanism of the cyanobacterial circadian clock.Methods Enzymol. 2022;666:59-78. doi: 10.1016/bs.mie.2022.02.011. Epub 2022 Mar 21. Methods Enzymol. 2022. PMID: 35465929
-
Studying RNA using site-directed spin-labeling and continuous-wave electron paramagnetic resonance spectroscopy.Methods Enzymol. 2009;469:303-28. doi: 10.1016/S0076-6879(09)69015-7. Epub 2009 Nov 17. Methods Enzymol. 2009. PMID: 20946796 Free PMC article.
Cited by
-
Hydrophilic Reduction-Resistant Spin Labels of Pyrrolidine and Pyrroline Series from 3,4-Bis-hydroxymethyl-2,2,5,5-tetraethylpyrrolidine-1-oxyl.Int J Mol Sci. 2024 Jan 26;25(3):1550. doi: 10.3390/ijms25031550. Int J Mol Sci. 2024. PMID: 38338825 Free PMC article.
-
In situ electron paramagnetic resonance spectroscopy using single nanodiamond sensors.Nat Commun. 2023 Oct 7;14(1):6278. doi: 10.1038/s41467-023-41903-5. Nat Commun. 2023. PMID: 37805509 Free PMC article.
-
Exploring protein structural ensembles: Integration of sparse experimental data from electron paramagnetic resonance spectroscopy with molecular modeling methods.Elife. 2024 Sep 16;13:e99770. doi: 10.7554/eLife.99770. Elife. 2024. PMID: 39283059 Free PMC article. Review.
-
Synthesis and Properties of (1R(S),5R(S),7R(S),8R(S))-1,8-Bis(hydroxymethyl)-6-azadispiro[4.1.4.2]tridecane-6-oxyl: Reduction-Resistant Spin Labels with High Spin Relaxation Times.Int J Mol Sci. 2023 Jul 15;24(14):11498. doi: 10.3390/ijms241411498. Int J Mol Sci. 2023. PMID: 37511257 Free PMC article.
-
Cracking the Code: Reprogramming the Genetic Script in Prokaryotes and Eukaryotes to Harness the Power of Noncanonical Amino Acids.Chem Rev. 2024 Sep 25;124(18):10281-10362. doi: 10.1021/acs.chemrev.3c00878. Epub 2024 Aug 9. Chem Rev. 2024. PMID: 39120726 Free PMC article. Review.
References
-
- Hubbell W. L. Altenbach C. Curr. Opin. Struct. Biol. 1994;4:566–573. doi: 10.1016/S0959-440X(94)90219-4. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

