Modulation of the antagonistic properties of an insulin mimetic peptide by disulfide bridge modifications
- PMID: 36633503
- PMCID: PMC10909431
- DOI: 10.1002/psc.3478
Modulation of the antagonistic properties of an insulin mimetic peptide by disulfide bridge modifications
Abstract
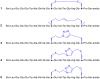
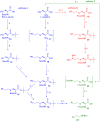
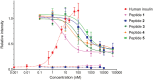
Insulin is a peptide responsible for regulating the metabolic homeostasis of the organism; it elicits its effects through binding to the transmembrane insulin receptor (IR). Insulin mimetics with agonistic or antagonistic effects toward the receptor are an exciting field of research and could find applications in treating diabetes or malignant diseases. We prepared five variants of a previously reported 20-amino acid insulin-mimicking peptide. These peptides differ from each other by the structure of the covalent bridge connecting positions 11 and 18. In addition to the peptide with a disulfide bridge, a derivative with a dicarba bridge and three derivatives with a 1,2,3-triazole differing from each other by the presence of sulfur or oxygen in their staples were prepared. The strongest binding to IR was exhibited by the peptide with a disulfide bridge. All other derivatives only weakly bound to IR, and a relationship between increasing bridge length and lower binding affinity can be inferred. Despite their nanomolar affinities, none of the prepared peptide mimetics was able to activate the insulin receptor even at high concentrations, but all mimetics were able to inhibit insulin-induced receptor activation. However, the receptor remained approximately 30% active even at the highest concentration of the agents; thus, the agents behave as partial antagonists. An interesting observation is that these mimetic peptides do not antagonize insulin action in proportion to their binding affinities. The compounds characterized in this study show that it is possible to modulate the functional properties of insulin receptor peptide ligands using disulfide mimetics.
Keywords: antagonism; dicarba; disulfide mimetics; insulin mimetic peptide; insulin receptor; staple; triazole.
© 2023 The Authors. Journal of Peptide Science published by European Peptide Society and John Wiley & Sons Ltd.
Figures

References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
