The Kaposi's sarcoma progenitor enigma: KSHV-induced MEndT-EndMT axis
- PMID: 36635149
- PMCID: PMC9957928
- DOI: 10.1016/j.molmed.2022.12.003
The Kaposi's sarcoma progenitor enigma: KSHV-induced MEndT-EndMT axis
Abstract
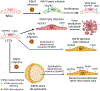
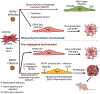
Endothelial-to-mesenchymal transition has been described in tumors as a source of mesenchymal stroma, while the reverse process has been proposed in tumor vasculogenesis and angiogenesis. A human oncogenic virus, Kaposi's sarcoma herpes virus (KSHV), can regulate both processes in order to transit through this transition 'boulevard' when infecting KS oncogenic progenitor cells. Endothelial or mesenchymal circulating progenitor cells can serve as KS oncogenic progenitors recruited by inflammatory cytokines because KSHV can reprogram one into the other through endothelial-to-mesenchymal and mesenchymal-to-endothelial transitions. Through these novel insights, the identity of the potential oncogenic progenitor of KS is revealed while gaining knowledge of the biology of the mesenchymal-endothelial differentiation axis and pointing to this axis as a therapeutic target in KS.
Keywords: Kaposi´s sarcoma herpesvirus; angiogenesis; endothelial-to-mesenchymal differentiation axis; sarcomagenesis; virus oncogenesis.
Copyright © 2022 The Author(s). Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of interests None are declared by the authors.
Figures

References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

