A TREM2-activating antibody with a blood-brain barrier transport vehicle enhances microglial metabolism in Alzheimer's disease models
- PMID: 36635496
- PMCID: PMC9991924
- DOI: 10.1038/s41593-022-01240-0
A TREM2-activating antibody with a blood-brain barrier transport vehicle enhances microglial metabolism in Alzheimer's disease models
Abstract
Loss-of-function variants of TREM2 are associated with increased risk of Alzheimer's disease (AD), suggesting that activation of this innate immune receptor may be a useful therapeutic strategy. Here we describe a high-affinity human TREM2-activating antibody engineered with a monovalent transferrin receptor (TfR) binding site, termed antibody transport vehicle (ATV), to facilitate blood-brain barrier transcytosis. Upon peripheral delivery in mice, ATV:TREM2 showed improved brain biodistribution and enhanced signaling compared to a standard anti-TREM2 antibody. In human induced pluripotent stem cell (iPSC)-derived microglia, ATV:TREM2 induced proliferation and improved mitochondrial metabolism. Single-cell RNA sequencing and morphometry revealed that ATV:TREM2 shifted microglia to metabolically responsive states, which were distinct from those induced by amyloid pathology. In an AD mouse model, ATV:TREM2 boosted brain microglial activity and glucose metabolism. Thus, ATV:TREM2 represents a promising approach to improve microglial function and treat brain hypometabolism found in patients with AD.
© 2023. The Author(s).
Conflict of interest statement
All authors, except P.M.Q., S.H., P.B., A.C., J.G., S.T.K., L.H.K., I.P., K.S., L.S., L.D.W., K.W.-M., M.R., L.M.B., M.B. and C.H., are full-time employees and/or shareholders of Denali Therapeutics. C.H. is a collaborator of Denali Therapeutics, participated on one advisory board meeting of Biogen and received a speaker honorarium from Novartis and Roche. C.H. is chief advisor of ISAR Bioscience and a member of the advisory board of AviadoBio. P.M.Q. is a full-time employee of Takeda Pharmaceuticals, Inc., a clinical development partner of Denali Therapeutics. This work has been described, in part, in pending patent applications.
Figures

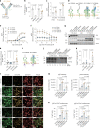








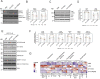

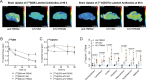
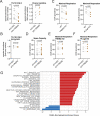
References
-
- Lewcock, J. W. et al. Emerging microglia biology defines novel therapeutic approaches for Alzheimer’s disease. Neuron108, 801–821 (2020). - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

