Cellular Uptake and Transport Mechanism of 6-Mercaptopurine Nanomedicines for Enhanced Oral Bioavailability
- PMID: 36636639
- PMCID: PMC9830076
- DOI: 10.2147/IJN.S394819
Cellular Uptake and Transport Mechanism of 6-Mercaptopurine Nanomedicines for Enhanced Oral Bioavailability
Abstract
Background: Nanomedicines have significant advantages in enhancing the oral bioavailability of drugs, but a deeper understanding of the underlying mechanisms remains to be interpreted. Hence, the present study aims to explain the uptake and trafficking mechanism for 6-MP nanomedicines we previously constructed.
Methods: 6-MP loaded poly(lactide-co-glycolide) (PLGA) nanomedicines (6-MPNs) were prepared by the multiple emulsion method. The transcytosis mechanism of 6-MPNs was investigated in Caco-2 cells, Caco-2 monolayers, follicle associated epithelium (FAE) monolayers and rats, including transmembrane pathway, intracellular trafficking, paracellular transport and the involvement of transporter.
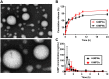
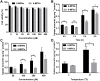
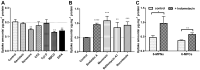
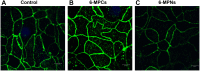
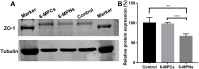
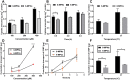
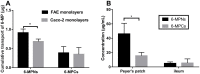
Results: Pharmacokinetics in rats showed that the area under the curve (AUC) of 6-MP in the 6-MPNs group (147.3 ± 42.89 μg/L·h) was significantly higher than that in the 6-MP suspensions (6-MPCs) group (70.31 ± 18.24 μg/L·h). The uptake of 6-MPNs in Caco-2 cells was time-, concentration- and energy-dependent. The endocytosis of intact 6-MPNs was mediated mainly through caveolae/lipid raft, caveolin and micropinocytosis. The intracellular trafficking of 6-MPNs was affected by endoplasmic reticulum (ER)-Golgi complexes, late endosome-lysosome and microtubules. The multidrug resistance associated protein 4 (MRP4) transporter-mediated transport of free 6-MP played a vital role on the transmembrane of 6-MPNs. The trafficking of 6-MPNs from the apical (AP) side to the basolateral (BL) side in Caco-2 monolayers was obviously improved. Besides, 6-MPNs affected the distribution and expression of zona occludens-1 (ZO-1). The transport of 6-MPNs in FAE monolayers was concentration- and energy-dependent, while reaching saturation over time. 6-MPNs improved the absorption of the intestinal Peyer's patches (PPs) in rats.
Conclusion: 6-MPNs improve the oral bioavailability through multiple pathways, including active transport, paracellular transport, lymphatic delivery and MRP4 transporter. The findings of current study may shed light on the cellular uptake and transcellular trafficking mechanism of oral nanomedicines.
Keywords: cellular uptake; nanomedicines; oral bioavailability; transport mechanism.
© 2023 Zou et al.
Conflict of interest statement
The authors declare that they have no competing interests in this work.
Figures
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

