Amygdala Intercalated Cells: Gate Keepers and Conveyors of Internal State to the Circuits of Emotion
- PMID: 36639901
- PMCID: PMC9761677
- DOI: 10.1523/JNEUROSCI.1176-22.2022
Amygdala Intercalated Cells: Gate Keepers and Conveyors of Internal State to the Circuits of Emotion
Abstract
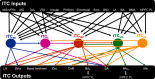
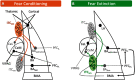
Generating adaptive behavioral responses to emotionally salient stimuli requires evaluation of complex associations between multiple sensations, the surrounding context, and current internal state. Neural circuits within the amygdala parse this emotional information, undergo synaptic plasticity to reflect learned associations, and evoke appropriate responses through their projections to the brain regions orchestrating these behaviors. Information flow within the amygdala is regulated by the intercalated cells (ITCs), which are densely packed clusters of GABAergic neurons that encircle the basolateral amygdala (BLA) and provide contextually relevant feedforward inhibition of amygdala nuclei, including the central and BLA. Emerging studies have begun to delineate the unique contribution of each ITC cluster and establish ITCs as key loci of plasticity in emotional learning. In this review, we summarize the known connectivity and function of individual ITC clusters and explore how different neuromodulators conveying internal state act via ITC gates to shape emotionally motivated behavior. We propose that the behavioral state-dependent function of ITCs, their unique genetic profile, and rich expression of neuromodulator receptors make them potential therapeutic targets for disorders, such as anxiety, schizophrenia spectrum, and addiction.
Copyright © 2022 the authors.
Figures


References
-
- Aggleton JP (1992) The amygdala: neurobiological aspects of emotion, memory, and mental dysfunction. New York: Wiley.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
