Opto-electromechanical quantification of epithelial barrier function in injured and healthy airway tissues
- PMID: 36644417
- PMCID: PMC9836726
- DOI: 10.1063/5.0123127
Opto-electromechanical quantification of epithelial barrier function in injured and healthy airway tissues
Abstract
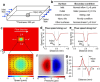
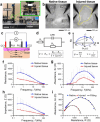
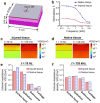
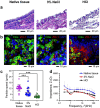
The airway epithelium lining the luminal surface of the respiratory tract creates a protective barrier that ensures maintenance of tissue homeostasis and prevention of respiratory diseases. The airway epithelium, unfortunately, is frequently injured by inhaled toxic materials, trauma, or medical procedures. Substantial or repeated airway epithelial injury can lead to dysregulated intrinsic repair pathways and aberrant tissue remodeling that can lead to dysfunctional airway epithelium. While disruption in the epithelial integrity is directly linked to degraded epithelial barrier function, the correlation between the structure and function of the airway epithelium remains elusive. In this study, we quantified the impact of acutely induced airway epithelium injury on disruption of the epithelial barrier functions. By monitoring alternation of the flow motions and tissue bioimpedance at local injury site, degradation of the epithelial functions, including mucociliary clearance and tight/adherens junction formation, were accurately determined with a high spatiotemporal resolution. Computational models that can simulate and predict the disruption of the mucociliary flow and airway tissue bioimpedance have been generated to assist interpretation of the experimental results. Collectively, findings of this study advance our knowledge of the structure-function relationships of the airway epithelium that can promote development of efficient and accurate diagnosis of airway tissue injury.
© 2023 Author(s).
Conflict of interest statement
The authors have no conflicts to disclose.
Figures



Similar articles
-
Diesel exhaust particle exposure exacerbates ciliary and epithelial barrier dysfunction in the multiciliated bronchial epithelium models.Ecotoxicol Environ Saf. 2024 Mar 15;273:116090. doi: 10.1016/j.ecoenv.2024.116090. Epub 2024 Feb 15. Ecotoxicol Environ Saf. 2024. PMID: 38364346
-
Regulation of airway immunity by epithelial miRNAs.Immunol Rev. 2021 Nov;304(1):141-153. doi: 10.1111/imr.13028. Epub 2021 Sep 21. Immunol Rev. 2021. PMID: 34549450 Free PMC article. Review.
-
Barrier function of airway tract epithelium.Tissue Barriers. 2013 Oct 1;1(4):e24997. doi: 10.4161/tisb.24997. Epub 2013 May 30. Tissue Barriers. 2013. PMID: 24665407 Free PMC article. Review.
-
Fluticasone Induces Epithelial Injury and Alters Barrier Function in Normal Subjects.J Steroids Horm Sci. 2014 Jun 11;5(2):26351. doi: 10.4172/2157-7536.1000134. J Steroids Horm Sci. 2014. PMID: 25324978 Free PMC article.
-
Rhinovirus Delays Cell Repolarization in a Model of Injured/Regenerating Human Airway Epithelium.Am J Respir Cell Mol Biol. 2016 Oct;55(4):487-499. doi: 10.1165/rcmb.2015-0243OC. Am J Respir Cell Mol Biol. 2016. PMID: 27119973 Free PMC article.
Cited by
-
A Minimally Invasive Robotic Tissue Palpation Device.IEEE Trans Biomed Eng. 2024 Jun;71(6):1958-1968. doi: 10.1109/TBME.2024.3357293. Epub 2024 May 20. IEEE Trans Biomed Eng. 2024. PMID: 38261510 Free PMC article.
-
Bioimpedance measurements of fibrotic and acutely injured lung tissues.Acta Biomater. 2025 Mar 1;194:270-287. doi: 10.1016/j.actbio.2025.01.039. Epub 2025 Jan 25. Acta Biomater. 2025. PMID: 39870150
References
Grants and funding
LinkOut - more resources
Full Text Sources

