Trajectories of depression among patients in treatment for opioid use disorder: A growth mixture model secondary analysis of the XBOT trial
- PMID: 36645265
- PMCID: PMC10332426
- DOI: 10.1111/ajad.13371
Trajectories of depression among patients in treatment for opioid use disorder: A growth mixture model secondary analysis of the XBOT trial
Abstract
Background and objectives: To inform clinical practice, we identified subgroups of adults based on levels of depression symptomatology over time during opioid use disorder (OUD) treatment.
Methods: Participants were 474 adults in a 24-week treatment trial for OUD. Depression symptoms were measured using the 17-item Hamilton Depression Rating Scale (HAM-D) at nine-time points. This was a secondary analysis of the Clinical Trials Network Extended-Release Naltrexone versus Buprenorphine for Opioid Treatment (XBOT) trial using a growth mixture model.
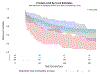
Results: Three distinct depression trajectories were identified: Class 1 High Recurring-10% with high HAM-D with initial partial reductions (of HAM-D across time), Class 2 Persistently High-5% with persistently high HAM-D, and Class 3 Low Declining-85% of the participants, with low HAM-D with early sustained reductions. The majority (low declining) had levels of depression that improved in the first 4 weeks and then stabilized across the treatment period. In contrast, 15% (high recurring and persistently high) had high initial levels that were more variable across time. The persistently high class had higher rates of opioid relapse.
Discussion and conclusions: In this OUD sample, most depressive symptomatology was mild and improved after medication treatment for opioid use disorder (MOUD). Smaller subgroups had higher depressive symptoms that persisted or recurred after the initiation of MOUD. Depressive symptoms should be followed in patients initiating treatment for OUD, and when persistent, should prompt further evaluation and consideration of antidepressant treatment.
Scientific significance: This study is the first to identify three distinct depression trajectories among a large clinical sample of individuals in MOUD treatment.
© 2023 The American Academy of Addiction Psychiatry (AAAP).
Conflict of interest statement
Declaration of Interest:
Marc Fishman has been a consultant for Alkermes, the manufacturer of XR-NTX. The authors alone are responsible for the content and writing of this paper.
Figures

Similar articles
-
Estimating the impact of stimulant use on initiation of buprenorphine and extended-release naltrexone in two clinical trials and real-world populations.Addict Sci Clin Pract. 2023 Feb 14;18(1):11. doi: 10.1186/s13722-023-00364-3. Addict Sci Clin Pract. 2023. PMID: 36788634 Free PMC article. Clinical Trial.
-
Longitudinal study of impact of medication for opioid use disorder on Hamilton Depression Rating Scale.J Affect Disord. 2022 Jan 15;297:148-155. doi: 10.1016/j.jad.2021.10.018. Epub 2021 Oct 17. J Affect Disord. 2022. PMID: 34670131 Clinical Trial.
-
Effectiveness of methadone versus buprenorphine in the treatment of opioid use disorder: secondary analyses of prospective cohort study data.BMJ Open. 2025 Jun 17;15(6):e095645. doi: 10.1136/bmjopen-2024-095645. BMJ Open. 2025. PMID: 40527567 Free PMC article.
-
Effects of buprenorphine on opioid craving in comparison to other medications for opioid use disorder: A systematic review of randomized controlled trials.Addict Behav. 2023 Apr;139:107589. doi: 10.1016/j.addbeh.2022.107589. Epub 2022 Dec 17. Addict Behav. 2023. PMID: 36565531
-
Sustained-release naltrexone for opioid dependence.Cochrane Database Syst Rev. 2025 May 9;5(5):CD006140. doi: 10.1002/14651858.CD006140.pub3. Cochrane Database Syst Rev. 2025. PMID: 40342086 Review.
Cited by
-
Suicidal ideation in adults with opioid use disorder treated with buprenorphine-naloxone versus extended-release naltrexone.Am J Drug Alcohol Abuse. 2025 Jul 11:1-9. doi: 10.1080/00952990.2025.2524110. Online ahead of print. Am J Drug Alcohol Abuse. 2025. PMID: 40643356
-
Cognitive behavioral therapy for anxiety and opioid use disorder: Development and pilot testing.J Subst Use Addict Treat. 2024 May;160:209296. doi: 10.1016/j.josat.2024.209296. Epub 2024 Jan 24. J Subst Use Addict Treat. 2024. PMID: 38272120 Free PMC article. Clinical Trial.
-
Exploring the impact of a medication adherence intervention on recovery and mental health outcomes: A secondary analysis of the MAT-PLUS pilot RCT.Drug Alcohol Depend Rep. 2025 May 16;15:100346. doi: 10.1016/j.dadr.2025.100346. eCollection 2025 Jun. Drug Alcohol Depend Rep. 2025. PMID: 40501493 Free PMC article.
References
-
- Center for Disease Control and Prevention. Drug Overdose Deaths. Published 2021. https://www.cdc.gov/drugoverdose/data/statedeaths.html
Publication types
MeSH terms
Substances
Grants and funding
- U10 DA013046/DA/NIDA NIH HHS/United States
- T32DA035165/DA/NIDA NIH HHS/United States
- HHSN271201200017C/DA/NIDA NIH HHS/United States
- U10 DA013714/DA/NIDA NIH HHS/United States
- K01 DA053391/DA/NIDA NIH HHS/United States
- UG1 DA013035/DA/NIDA NIH HHS/United States
- K01DA053391/DA/NIDA NIH HHS/United States
- K24 DA022412/DA/NIDA NIH HHS/United States
- U10 DA015833/DA/NIDA NIH HHS/United States
- U10 DA013720/DA/NIDA NIH HHS/United States
- T32 DA035165/DA/NIDA NIH HHS/United States
- U10 DA015831/DA/NIDA NIH HHS/United States
- U10 DA013035/DA/NIDA NIH HHS/United States
- HHSN271201500065C/DA/NIDA NIH HHS/United States
- U10 DA013732/DA/NIDA NIH HHS/United States
- L30 DA056944/DA/NIDA NIH HHS/United States
- UG1DA013035/DA/NIDA NIH HHS/United States
- U10 DA013045/DA/NIDA NIH HHS/United States
- U10 DA013034/DA/NIDA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical

