Reactive oxygen species in status epilepticus
- PMID: 36648377
- PMCID: PMC10173846
- DOI: 10.1002/epi4.12691
Reactive oxygen species in status epilepticus
Abstract
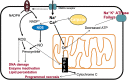
It has long been recognized that status epilepticus can cause considerable neuronal damage, and this has become one of its defining features. The mechanisms underlying this damage are less clear. Excessive activation of NMDA receptors results in large rises in internal calcium, which eventually lead to neuronal death. Between NMDA receptor activation and neuronal death are a number of intermediary steps, key among which is the generation of free radicals and reactive oxygen and nitrogen species. Although it has long been thought that mitochondria are the primary source for reactive oxygen species, more recent evidence has pointed to a prominent role of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, an enzyme localized in cell membranes. There is burgeoning in vivo and in vitro evidence that therapies that target the production or removal of reactive oxygen species are not only effective neuroprotectants following status epilepticus, but also potently antiepileptogenic. Moreover, combining therapies targeted at inhibiting NADPH oxidase and at increasing endogenous antioxidants seems to offer the greatest benefits.
Keywords: excitotoxicity; free radicals; mitochondria; reactive oxygen species; status epilepticus.
© 2023 The Author. Epilepsia Open published by Wiley Periodicals LLC on behalf of International League Against Epilepsy.
Conflict of interest statement
MW has no conflict of interest to disclose.
Figures

Similar articles
-
Reactive oxygen species in status epilepticus.Epilepsy Behav. 2019 Dec;101(Pt B):106410. doi: 10.1016/j.yebeh.2019.07.011. Epub 2019 Aug 1. Epilepsy Behav. 2019. PMID: 31378559 Review.
-
Seizure activity results in calcium- and mitochondria-independent ROS production via NADPH and xanthine oxidase activation.Cell Death Dis. 2014 Oct 2;5(10):e1442. doi: 10.1038/cddis.2014.390. Cell Death Dis. 2014. PMID: 25275601 Free PMC article.
-
Pharmacological strategies to lower crosstalk between nicotinamide adenine dinucleotide phosphate (NADPH) oxidase and mitochondria.Biomed Pharmacother. 2019 Mar;111:1478-1498. doi: 10.1016/j.biopha.2018.11.128. Epub 2019 Feb 14. Biomed Pharmacother. 2019. PMID: 30841463 Review.
-
MicroRNA Targeting Nicotinamide Adenine Dinucleotide Phosphate Oxidases in Cancer.Antioxid Redox Signal. 2020 Feb 10;32(5):267-284. doi: 10.1089/ars.2019.7918. Epub 2019 Nov 21. Antioxid Redox Signal. 2020. PMID: 31656079 Review.
-
Baccharis trimera inhibits reactive oxygen species production through PKC and down-regulation p47 phox phosphorylation of NADPH oxidase in SK Hep-1 cells.Exp Biol Med (Maywood). 2017 Feb;242(3):333-343. doi: 10.1177/1535370216672749. Epub 2016 Oct 7. Exp Biol Med (Maywood). 2017. PMID: 28103717 Free PMC article.
Cited by
-
Peripheral and central effects of NADPH oxidase inhibitor, mitoapocynin, in a rat model of diisopropylfluorophosphate (DFP) toxicity.Front Cell Neurosci. 2023 Jun 21;17:1195843. doi: 10.3389/fncel.2023.1195843. eCollection 2023. Front Cell Neurosci. 2023. PMID: 37416507 Free PMC article.
-
Association between composite dietary antioxidant index and epilepsy in American population: a cross-sectional study from NHANES.BMC Public Health. 2024 Aug 17;24(1):2240. doi: 10.1186/s12889-024-19794-y. BMC Public Health. 2024. PMID: 39154181 Free PMC article.
-
Ferroptosis and epilepsy: bidirectional pathogenic links and therapeutic implications.Front Neurol. 2025 Jul 30;16:1635441. doi: 10.3389/fneur.2025.1635441. eCollection 2025. Front Neurol. 2025. PMID: 40808923 Free PMC article. Review.
-
Beneficial Effects of Rosmarinic Acid In Vitro and In Vivo Models of Epileptiform Activity Induced by Pilocarpine.Brain Sci. 2023 Feb 8;13(2):289. doi: 10.3390/brainsci13020289. Brain Sci. 2023. PMID: 36831832 Free PMC article.
-
Oxidative Stress and Neurodegeneration in Animal Models of Seizures and Epilepsy.Antioxidants (Basel). 2023 May 5;12(5):1049. doi: 10.3390/antiox12051049. Antioxidants (Basel). 2023. PMID: 37237916 Free PMC article. Review.
References
-
- Wasterlain CG. Breakdown of brain polysomes in status epilepticus. Brain Res. 1972;39(1):278–84. - PubMed
-
- Meldrum BS, Brierley JB. Prolonged epileptic seizures in primates. Ischemic cell change and its relation to ictal physiological events. Arch Neurol. 1973;28(1):10–7. - PubMed
-
- Meldrum BS, Horton RW. Physiology of status epilepticus in primates. Arch Neurol. 1973;28(1):1–9. - PubMed
-
- Lipton SA, Rosenberg PA. Excitatory amino acids as a final common pathway for neurologic disorders. N Engl J Med. 1994;330(9):613–22. - PubMed
-
- Tanaka H, Grooms SY, Bennett MV, Zukin RS. The AMPAR subunit GluR2: still front and center‐stage. Brain Res. 2000;886(1–2):190–207. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

