Broad-scope Syntheses of [11 C/18 F]Trifluoromethylarenes from Aryl(mesityl)iodonium Salts
- PMID: 36652272
- PMCID: PMC10192059
- DOI: 10.1002/chem.202204004
Broad-scope Syntheses of [11 C/18 F]Trifluoromethylarenes from Aryl(mesityl)iodonium Salts
Abstract
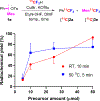
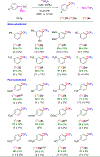
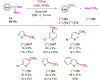
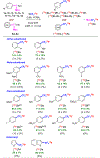
Efficient methods for labeling aryl trifluoromethyl groups to provide novel radiotracers for use in biomedical research with positron emission tomography (PET) are keenly sought. We report a broad-scope method for labeling trifluoromethylarenes with either carbon-11 (t1/2 =20.4 min) or fluorine-18 (t1/2 =109.8 min) from readily accessible aryl(mesityl)iodonium salts. In this method, the aryl(mesityl)iodonium salt is treated rapidly with no-carrier-added [11 C]CuCF3 or [18 F]CuCF3 . The mesityl group acts as a spectator allowing radiolabeled trifluoromethylarenes to be obtained with very high chemoselectivity. Radiochemical yields from aryl(mesityl)iodonium salts bearing either electron-donating or electron-withdrawing groups at meta- or para- position are good to excellent (67-96 %). Ortho-substituted and otherwise sterically hindered trifluoromethylarenes still give good yields (15-34 %). Substituted heteroaryl(mesityl)iodonium salts are also viable substrates. The broad scope of this method was further exemplified by labeling a previously inaccessible target, [11 C]p-trifluoromethylphenyl boronic acid, as a potentially useful labeling synthon. In addition, fluoxetine, leflunomide, and 3-trifluoromethyl-4-aminopyridine, as examples of small drug-like molecules and candidate PET radioligands, were successfully labeled in high yields (69-81 %).
Keywords: PET radiochemistry; carbon-11; diaryliodonium salts; fluorine-18; trifluoromethyl.
© 2023 Wiley-VCH GmbH.
Conflict of interest statement
CONFLICT OF INTEREST
The authors declare no competing conflict of interest.
Figures





References
-
- Willmann JK, van Bruggen N, Dinkelborg LM, Gambhir SS, Nat. Rev. Drug. Discov. 2008, 7, 591–607; - PubMed
- Donnelly DJ, Sem. Nucl. Med. 2017, 47, 454–460; - PubMed
- Suridjan I, Comley RA, Rabiner EA, Brain Imaging Behav. 2019, 13, 354–365; - PubMed
- Donnelly DJ, Ch. 22, pp. 703–725, in Handbook of Radiopharmaceuticals, 2nd Ed., Kilbourn MR, Scott PJ (Eds), 2021. John Wiley & Sons Ltd.;
- Ghosh KK, Padmanabhan P, Yang C-T, Ng DCE, Palanivel M, Mishra S, Halldin C, Gulyás B Drug Discov. Today, 2022, 27, 280–291. - PubMed
-
- Pruis IJ, van Dongen GAMS, Veldhuijzen van Zanten SEM, Int. J. Mol. Sci. 2020, 21, 1029; - PMC - PubMed
- Unterrainer M, Eze, Ilhan H, Marschner S, Roengvoraphoj O, Schmidt-Hegemann NS, Walter F, Kunz WG, Munck af Rosenschöld P, Jeraj R, Albert NL, Grosu AL, Niyazi M, Bartenstein P, Belka C Radiat. Oncol. 2020, 15, 88. - PMC - PubMed
-
- https://www.xemed.com/markets/, Last accessed 4th March 2021.
-
- Yale HL, J. Med. Pharm. Chem. 1958, 1, 121–133; - PubMed
- Purser S, Moore PR, Swallow S, Gouverneur V, Chem. Soc. Rev. 2008, 37, 320–330; - PubMed
- Hagmann WK, J. Med. Chem. 2008, 51, 4359–4369; - PubMed
- Zhu W, Wang J, Wang S, Gu Z, Aceña JL, Izawa K, Liu H, Soloshonok VA, J. Fluorine Chem. 2014, 167, 37–54;
- Gillis EP, Eastman KJ, Hill MD, Donnelly DJ, Meanwell NA, J. Med. Chem. 2015, 58, 8315–8359; - PubMed
- Xu Z, Zhu Z, Peng C, Chen Z, Zhu W, Aisa HA, J. Chem. Inf. Model. 2020, 60, 6242–6250; - PubMed
- Francis F, Wuest F, Molecules 2021, 26, 6478. - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

