Intralesional Corticosteroid Administration in the Treatment of Keloids: A Scoping Review on Injection Methods
- PMID: 36657423
- PMCID: PMC10906477
- DOI: 10.1159/000529220
Intralesional Corticosteroid Administration in the Treatment of Keloids: A Scoping Review on Injection Methods
Abstract
Background: Intralesional corticosteroid administration (ICA) is a first-line treatment for keloids. However, its clinical results are still highly variable and often suboptimal. Treatment results may strongly be influenced by various operator-dependent factors. The aim of this study was to map the details of ICA in keloids described in randomized controlled trials (RCTs), hence presenting the scientific practice of a first-line treatment for keloids in the best available evidence.
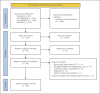
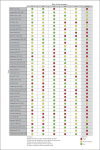
Summary: A systematic search was performed on PubMed, Ovid MEDLINE, Ovid EMBASE, and CENTRAL. Eligible studies were RCTs including patients with keloids treated with intralesional corticosteroids. Treatment and study design-related data were charted on a predefined form. Thirty-eight RCTs were included for data extraction. Triamcinolone acetonide was used in 37 (97.4%) studies. Dosing per cm2 could only be compared among ten (26%) studies and varied from 1 to 20 mg. The maximum dose per session varied from 20 to 80 mg. Local anesthetics were administered in seven (20%) RCTs. Treatment intervals varied from weekly to monthly, with 4 weeks most frequently (50%) used. Needle size was reported in eleven (29%) studies and varied from 26 to 30-gauge. Syringe size was specified in four (11%) studies, being 1 mL. The injection level was described in eleven (29%) studies. Blanching as endpoint was reported in ten (26%) studies. Outcome measures varied widely, from height, surface area, or volume, to Vancouver Scar Scale, Patient and Observer Scar Assessment Scale, pain and itch scores, patient satisfaction, and different efficacy rates. Only six studies had a follow-up of ≥6 months. Recurrence was identified in two studies with 18 weeks and 1 year of follow-up. Adverse events were reported in 23 (61%) studies.
Keywords: Corticosteroid; Injection; Intralesional corticosteroids; Keloid; Triamcinolone.
© 2023 The Author(s). Published by S. Karger AG, Basel.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures

References
-
- Furtado F, Hochman B, Ferrara SF, Dini GM, Nunes JMC, Juliano Y, et al. What factors affect the quality of life of patients with keloids? Rev Assoc Med Bras. 2009;55((6)):700–704. - PubMed
-
- Bijlard E, Kouwenberg CAE, Timman R, Hovius SER, Busschbach JJV, Mureau MAM. Burden of keloid disease a cross-sectional health-related quality of life assessment. Acta Derm Venereol. 2017;97((2)):225–229. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

